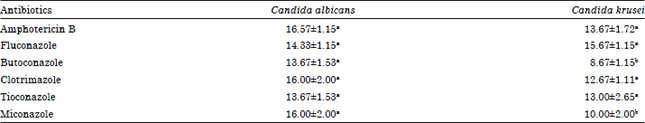
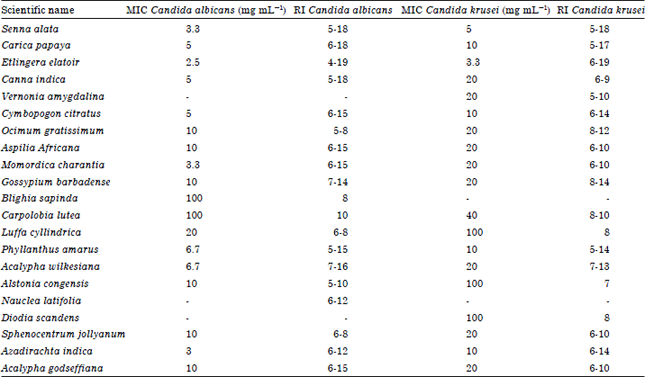
Research Article
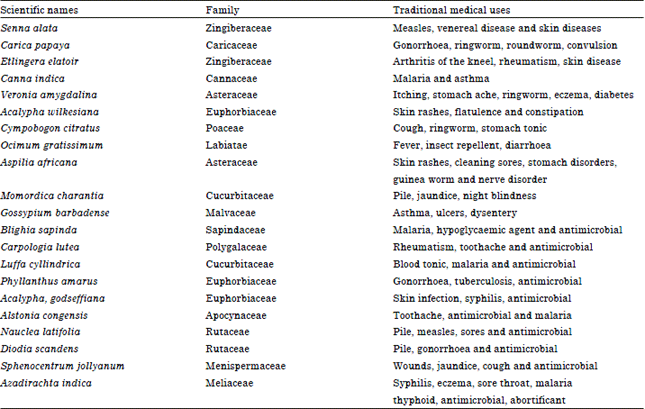
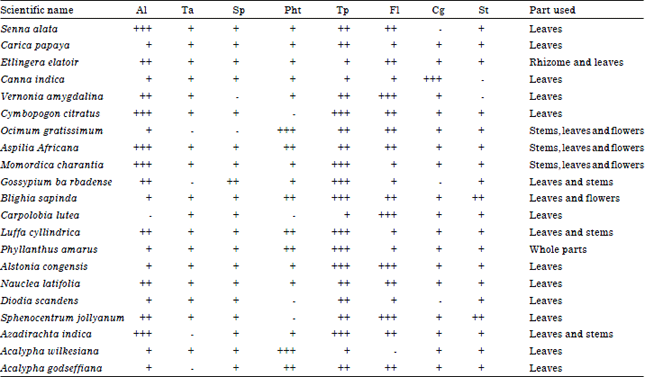
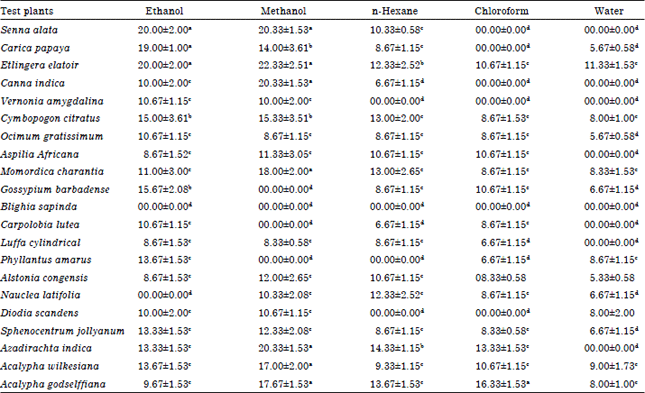
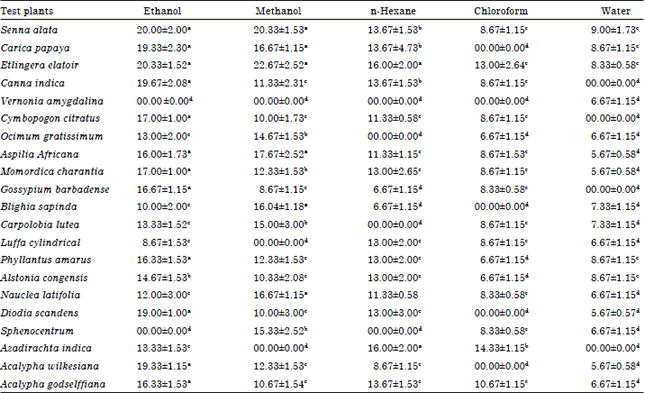
Screening of Some Nigerian Medicinal Plants for Anti-candida Activity
Department of Botany, University of Lagos, Akoka, Yaba, Lagos, Nigeria
LiveDNA: 234.38355
Egboduku O. Wisdom
Department of Botany, Delta State University, Abraka, Delta State, Nigeria