ABSTRACT
Plant biotechnology is an attractive means for improving cotton, its use requires an efficient regeneration system from somatic tissues of cotton plants. Cotton is an important economic and fibre crop, grown in 70 countries in the world. Somatic embryogenesis and plant regeneration are fundamental to the genetic improvement of cotton using biotechnology and genetic transformation. Browning and subsequent death of the cultured explants are major problems for many tissue culture systems. In this study, different explants and various concentrations of PGRs were tested in order to minimize excretion of phenolic compound and their inhibitory effects in cotton tissue culture system. Cotyledons and meristematic shoot tips had the minimal excretion of phenolic compound, also in medium with 27.12 μM 2,4-D and 8.87 μM BA phenolic compound was the least. Meristematic shoot tips in medium with 0.492 μM IBA and 0.929 μM KIN regenerated whole plantlets without any excretion phenolic compound. Therefor, in this study inhibitory effects due to excretion of phenolic compound in tissue culture media were minimum.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajdb.2010.16.22
URL: https://scialert.net/abstract/?doi=ajdb.2010.16.22
INTRODUCTION
Cotton is an economically important crop that is grown throughout the world. It is grown as a source of fibre, food and feed Lint, the most economically important product from the cotton plant, provides a source of high quality fibre of the textile industry. Cotton seed is an important source of oil and cotton seed meal is a high protein product used as livestock feed (Keshamma et al., 2008). Although, great progress has been made in the field of improvement of cotton with conventional breeding methodology, it is time-consuming and commercialization of new cotton varieties often takes 6 to 10 years. Compatibility limitations narrow the gene pool available for this process.
A number of these shortcomings may be overcome by plant biotechnology. For example, control can be exerted over selection of the gene(s) and its expression. The gene pool can be expanded to all living organisms (plants, animals, bacteria and fungi). Researcher notes that as technology is refined, custom-made synthetic genes will become another source for desired traits (Zhang et al., 2000). Thus, cotton biotechnology can be significantly applied for the improvement of cotton. Although, plant biotechnology is an attractive means for improving cotton, its use requires an effective regeneration system from somatic tissues of cotton plants (Zhang et al., 2001).
One of the major problems for many tissue culture system is browning and subsequent death of the cultured explants that usually depended on the phenolic compounds and the quantity of total phenols Ozyigit (2008).
Phenolic compounds occur as secondary metabolites in all plant species and they are generally characterized by a benzene ring and one hydroxyl group (Antolovich et al., 2000; Kefeli et al., 2003). They are also extremely diverse compounds, for example carnosol and rosmanol, which are diterpenes were identified in herbs and spices while the main phenolics are isoflavone glycosides and several phenolic acids like ferulic, caffeic and chlorogenic acids which are present in soybean (Robards et al., 1999). Plant phenols are classified into major groupings distinguished by the number of constitutive carbon atoms in conjunction with the structure of basic phenolic skeleton (Robards et al., 1999; Antolovich et al., 2000). Shikimate is the starting product for the biosynthesis of most phenolic compounds. They are also acidic substances, due to the dissociability of their -OH groups. Many phenols are rather reactive compounds and as long as no steric inhibition due to additional side chains occurs, they form hydrogen bonds. The composition and synthesis of phenolics in plant tissues may determine by genetic and environmental conditions like oxidative reactions during culturing, processing and storage (Lux-Endrich et al., 2000). It seems that there is a relation between chemical compound of media and phenolic exudation, media discoloration, rooting deficiencies and explant browning and death. For example plant phenolics are modulators of Indole Acetic Acid (IAA) catabolism. Some monophenols like synaptic acid and ferulic acid, at low concentrations, inhibit enzymatic oxidation of IAA and this results in cell elongation and cell division and subsequent plant growth and development (Volpert et al., 1995; Arnaldos et al., 2001). It was noticed that plant phenolics increase the rigidity of plant cell walls acting as molecular bridges between cell wall components (Ozyigit, 2008).
In this study effect of plant growth regulators for organ formation and phenolic compound in cotton tissue culture media were examined.
MATERIALS AND METHODS
This study conducted in Iranian Research Institute of Plant Protection from Aug. 2008 to Sep. 2009
Seed Germination and Culture of Aseptic Seedlings
Mature seeds of cotton (Gossypium hirsutum L.) were surface sterilized by commercial bleach, ethanol 70%, flame of an alcohol burner for a moment, sterile distilled water, tap water, H2O2 and sterile filter papers. The surface sterilized seeds were germinated on MS Murashige and Skoog (1962) medium. Medium supplemented with 1% agar and 3% sucrose for germination at 25±2°C under 24 h photoperiod conditions with the light intensity of approximately 2000 Lux.
Induction and Proliferation of Callus and Organogenesis
Hypocotyl sections (6-9 mm), cotyledon pieces (10-16 mm 2) area, root segments (4-6 mm) and meristematic shoot tips (1-2 mm) of 7 days old sterile seedlings were placed on MS medium supplemented with various concentrations of Plant Growth Regulators (PGRs) (BA, IBA, 2,4-D and KIN) for the induction of callus and organogenesis. Cultures were maintained in 24 h photoperiod conditions with the light intensity of approximately 2000 Lux at 25±2°C. After establishment, cultures were subcultured at 5-6 weeks intervals on fresh media and 2 months later the means number of browned and died cultured explants in different media were counted.
Experimental Design, Data Collection and Analysis
Experiments were set up in Completely Randomized Design and repeated four times. Each treatment has 16 replications. Observation on the browned or died explants and the Number of explants with callus or organ were recorded. Data were subjected to SD and ANOVA test.
RESULTS
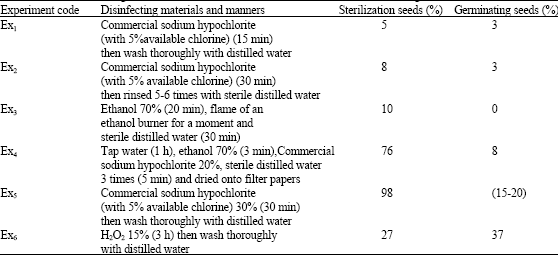
Different seeds and concentrations of disinfecting material were used in this experiments for establishment of sterile seeds cotton. Percentage of sterilization and germination of seeds varied in different manners. These variation are shown in Table 1. In Ex5 that commercial sodium hypochlorite 30% were used for 30 min cotton seeds were establishment in the best way.
In this study various explants and media for induction and growth of calli, organogenesis, direct regeneration response and effect of PGRs compound and amounts of phenolic compound were investigated. Present studies showed that either various explants or different media are very effective (Table 2). All of root explants on all of media browned and died due to excretion and oxidation of phenolic compound.
| Table 1: | Sterilization and germination of cotton cv varamin seeds with various disinfecting materials and manners |
 | |
| Table 2: | Callus induction and organogenesis of cotton Gossypium hirsutum L. variety varamin from different explants and different concentrations of PGRs (μM L-1) |
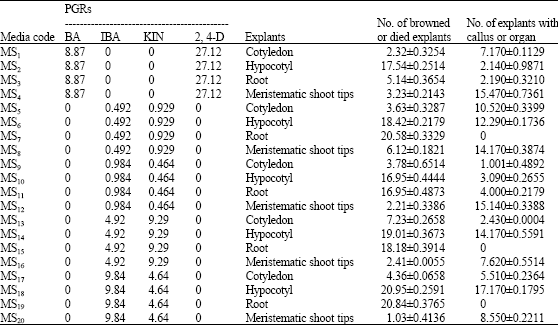 | |
| Data represent means of 16 replicates. There is significant difference between treatment and control at p<0.01 | |
Cotyledon explants on media with 0.492 μM IBA+0.929 μM KIN, 0.984 μM IBA+0.464 μM KIN, 4.92 μM IBA+9.29 μM KIN, 9.84 μM IBA+4.64 μM KIN, did not synthesize phenolic compound. In this media cotyledon explants at first growth and increased their diameters then in medium with 0.492 μM IBA + 0.928 μM KIN induced callus. Growth of calli in this conditions was very slow (Fig. 1).
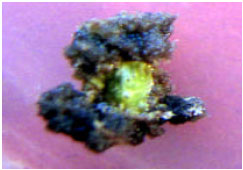
Response of hypocotyl in different media was very various. This variation related to PGRs content of media. In media with KIN and IBA callus was induced and growth. These calli were two kinds. Organoge and nonorganogen. In organogene calli when diameter of calli increased to 5-6 mm induced roots. These roots was white, either length or thickness of them growth very well and browned in media (Fig. 2). Some of calli in these media only growth and browned severity (Fig. 3). Also in these media regenerated roots and regenerated leaves were induced on one hypocotyl explants (Fig. 4).
 | |
| Fig. 1: | Callusing on cotyledon explants in 0.492μM IBA and 0.929 μM KIN |
 | |
| Fig. 2: | Callusing and rooting on hypocotyl explants in 0.492 μM IBA and 0.929 μM KIN |
 | |
| Fig. 3: | Callusing on hypocotyl explants in 9.84 μM IBA and 4.64 μM KIN |
 | |
| Fig. 4: | Regenerations of leaves on hypocotyl explants in 0.984 μM IBA and 0.464 μM KIN |
 | |
| Fig. 5: | Callusing on hypocotyl explants in 27.12 μM 2, 4-D and 8.87 μM BA |
 | |
| Fig. 6: | Regeneration of four week old plantlets on meristematic shoot tip in 0.492 μM IBA and 0.929 μM KIN |
In medium with 27.12 μM 2, 4-D and 2 8.87 μM BA severity of phenolic compound excretion was very slow and nearly was zero (Fig. 5). On hypocotyl explants in this medium calli induced and growth very well (Fig. 5).
As could been seen in Fig. 3 and 5 these calli that induced on hypocotyl explants had two important differences due to explant polarity for inducing of calli (polar and bipolar).
Meristematic shoot tips in media induced regenerated plants without excretion of phenolic compound abundantly. In Fig. 6 four week old regenerated plantlets on MS medium supplemented with 0.492 μM IBA and 0.929 μM KIN were seen.
DISCUSSION
There are many methods for surface sterilization of cotton seed (Zhang et al., 2001), Ozyigit (2008), Ikram-ul-Haq (2005), (Zhang et al., 2003). All of these methods were tested in this experiments but use of commercial bleach (30%) for 30 min were suitable for Iranian cotton seed, it is clear that natural conditions and normal floral of Iranian field are effective.
In this study different explants and media were used and were confirmed that amount of phenolic compound and their inhibitor effectives could be varied. Volpert et al. (1995) and Arnaldos et al. (2001) declared that plant phenolics are modulators of Indole Acetic Acid (IAA) catabolism. Some monophenols like synaptic acid and ferulic acid, at low concentrations, inhibit enzymatic oxidation of IAA and this results in cell elongation and cell division and subsequent plant growth and development.
As it is known, phenolics are synthesized in leaves and then carried to other tissues and organs. Therefore, amounts of total phenolic compounds in leaves are more than the other tissues and organs of the plants (Ozyigit, 2008). But in this study phenolic compounds are synthesized in roots was more than to other explants, also phenolic compounds were synthesized in leaves were less to other explants.
As was noticed above composition of media influenced on total phenolic compounds and excretion of them into media also. Hypocotyl explants in medium with 27.12 μM 2,4-D and 8.8 μM BA had the least production of phenolic compound. These explants in medium 0.492 μM IBA and 0.929 μM KIN browned severely (Fig. 3). Therefore, we can decrease inhibitor effectives of phenolic compound in cotton tissue culture with election of suitable explants and suitable media.
Meristematic shoot tips in MS medium plus 0.492 IBA μM and 0.929 μM KIN regenerated whole plantlets. On the other hand, these explants had very little phenolic compounds. This resulted in research of Ozyigit (2008) with 4.64 μM KIN.
In medium supplemented by 27.12 μM 2,4-D and 8.87 μM BA hypocotyl explants induced and growth of calli at the highest level, whereas the calli induced and growth very well in 0.57 μM IAA, 0.452 μM 2,4-D and 0.464 μM KIN (Zhang et al., 2003).
Zhang et al. (2001) stated that for callus inducing in cotton the best explants are hypocotyls. Also, in this study hypocotyls explants induced calli with powerful growth.
As were explained earlier meristematic shoot tips in MS medium plus 0.492 μM IBA and 0.929 μM KIN regenerated whole plantlets. Also expressed that meristematic shoot tips had low level of phenolic compound in media.
Based on our results the most important factors for reduction of phenolic compound in tissue culture media are composition of media and type of explants, whereas there are researchers that believed another factors such as age of explants and genotype are the most effective factors for total amount of phenolic compound in media (Ozyigit et al., 2007; Gupta et al., 2000; Kumria et al., 2003; Lorenzo and Angeles, 2001).
In this research we report that could been overcome phenolic compound excretion in tissue culture media simply by variation of explants and PGRs for callusing and organogenesis without to need addition of external material such as activated charcoal Wang and Huang (2009).
REFERENCES
- Arnaldos, T.L., R. Munoz, M.A. Ferrer and A.A. Calderon, 2001. Changes in phenol content during strawberry (Fragaria x ananasa, cv. Chandler) callus culture. Physiologia Plantarum, 113: 315-322.
CrossRefDirect Link - Gupta, S.K., P.K. Singh, S.V. Sawant, R. Chaturvedi and R. Tuli, 2000. Effect of light intensity on in vitro multiple shoot induction and regeneration of cotton (Gossypium hirsutum L. cv Khandawa-2). Indian J. Exp. Biol., 38: 399-401.
PubMed - Ikram-ul-Haq, 2005. Callus proliferation and somatic embryogenesis in cotton (Gossypium hirsutum L.). Afr. J. Biotechnol., 4: 206-209.
Direct Link - Kefeli, V.I., M.V. Kalevitch and B. Borsari, 2003. Phenolic cycle in plants and environment. J. Cell Mol. Biol., 2: 13-18.
Direct Link - Keshamma, E., S. Rohini, K.S. Rao, B. Madhusudhan and M.U. Kumar, 2008. Molecular biology and physiology. J. Cotton Sci., 12: 264-272.
Direct Link - Kumria, R., V.G. Sunnichan, D.K. Das, S.K. Gupta, V.S. Reddy, R.K. Bhatnagar and S. Leekavathi, 2003. High-frequency somatic embryo production and maturation into normal plants in cotton (Gossypium hirsutum L.) through metabolic stress. Plant Cell Rep., 21: 635-639.
CrossRef - Lorenzo, J.C., M. de los Angeles Blanco, O. Pelaez, A. Gonzalez and M. Cid et al., 2001. Sugarcane microporopogation and phenolic excretion. Plant Cell Tissue Organ Cult., 65: 1-8.
CrossRef - Lux-Endrich, A., D. Treutter and W. Feucht, 2000. Influence of nutrients and carbohydrate supply on the phenol composition of apple shoot cultures. Plant Cell Tissue Organ Cult., 60: 15-21.
CrossRef - Murashige, T. and F. Skoog, 1962. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant., 15: 473-497.
CrossRefDirect Link - Ozyigit, I.I., 2008. Phenolic changes during in vitro organogenesis of cotton (Gossypium hirsutum L.) shoot tips. Afr. J. Biotechnol., 7: 1145-1150.
Direct Link - Ozyigit, I.I., M.V. Kahraman and O. Ercan, 2007. Relation between explant age, total phenols and regeneration response in tissue cultured cotton (Gossypium hirsutum L.). Afr. J. Biotechnol., 6: 3-8.
Direct Link - Robards, K., P.D. Prenzeler, G. Tucker, P. Swatsitang and W. Glover, 1999. Phenolic compounds and their role in oxidative processes in fruits. Food Chem., 66: 401-436.
CrossRefDirect Link - Volpert, R., W. Osswald and E.F. Elstner, 1995. Effects of cinnamic acid derivates on indole acetic acid oxidation by peroxidase. Phytochemistry, 38: 19-22.
CrossRef - Wang, P. and L. Huang, 2009. Beneficial effects of activated charcoal on plant tissue and organ culture. In vitro Cell. Dev. Biol. Plant, 12: 260-262.
CrossRefDirect Link - Li, Y.Z., X.H. Zheng, H.L. Tang, J.W. Zhu and J.M. Yang, 2003. Increase of β-1, 3-glucanase and chitinase activities in cotton callus cells treated by salicylic acid and toxin of Verticillium dahliae. Acta Bot. Sin., 45: 802-808.
Direct Link - Zhang, B.H., F. Liu, C.B. Yao and K.B. Wang, 2000. Recent progress in cotton biotechnology and genetic engineering in China. Curr. Sci., 79: 37-44.
Direct Link - Zhang, B.H., Q.L. Wang and F. Liu, 2001. Phenotypic variation in cotton (Gossypium hirsutum L.) regenerated plants. Curr. Sci., 8: 1112-1115.
Direct Link - Antolovich, M., P.D. Prenzler, K. Robards and D. Ryan, 2000. Sample preparation in the determination of phenolic compounds in fruits. Analyst, 125: 989-1009.
CrossRefDirect Link








