Research Article
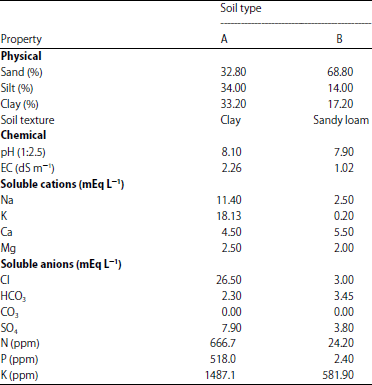
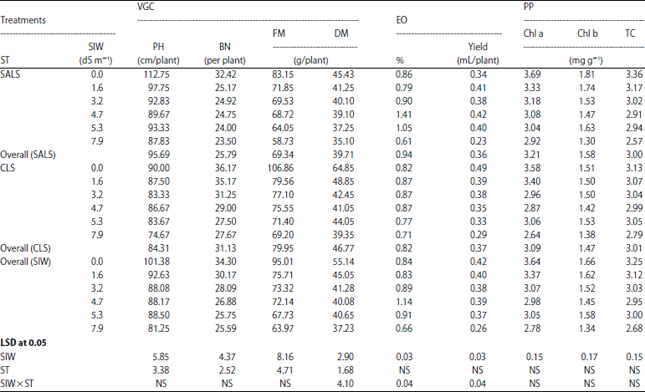
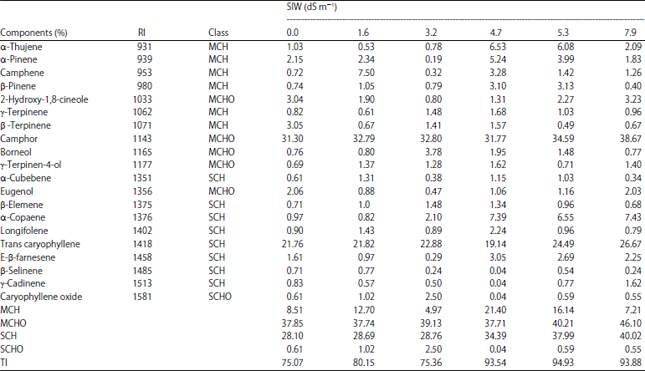
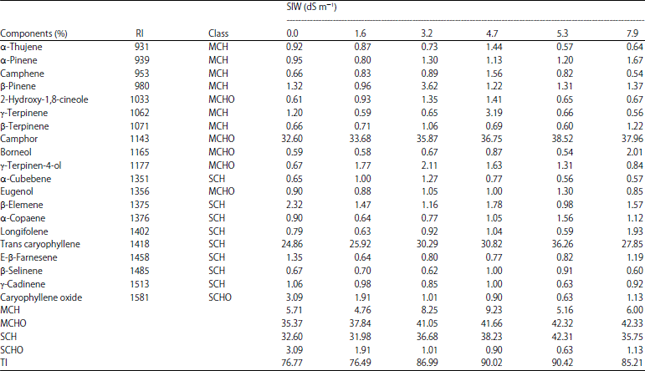
Effect of Salinity Stress and Soil Types on Growth, Photosynthetic Pigments and Essential Oil of Artemisia annua L.
Department of Ornamental Horticulture, Faculty of Agriculture, Cairo University, Giza, Egypt
Khalid Ali Khalid
Department of Medicinal and Aromatic Plants, National Research Centre, 12311 Dokki, Cairo, Egypt
LiveDNA: 20.3160
Amaal Abd-Elkhalek Mohamed Heikal
Department of Ornamental Horticulture, Faculty of Agriculture, Cairo University, Giza, Egypt
Mohammad Mahmoud Nagdy
Department of Medicinal and Aromatic Plants, National Research Centre, 12311 Dokki, Cairo, Egypt