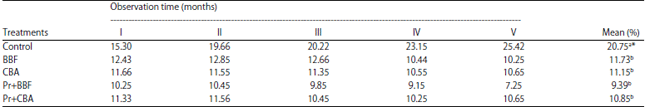
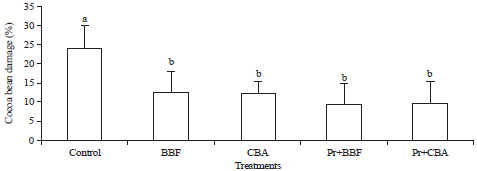
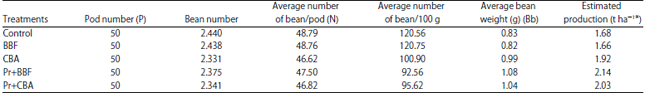
Research Article
Applications of Biological Agents and Pruning Effectively Control Cocoa Pod Borer
Department of Plant Pests and Diseases, Faculty of Agriculture, Tadulako University Palu, 94118 Central Sulawesi, Indonesia
LiveDNA: 62.19768
Muhammad Basir-Cyio
Department of Agroecotechnology, Faculty of Agriculture, Tadulako University, Palu, 94118 Central Sulawesi, Indonesia
Uswah Hasanah
Department of Agroecotechnology, Faculty of Agriculture, Tadulako University, Palu, 94118 Central Sulawesi, Indonesia
Mahfudz
Department of Agroecotechnology, Faculty of Agriculture, Tadulako University, Palu, 94118 Central Sulawesi, Indonesia
Shahabuddin Saleh
Department of Plant Pests and Diseases, Faculty of Agriculture, Tadulako University Palu, 94118 Central Sulawesi, Indonesia
LiveDNA: 62.20200
Nur Edy
Department of Plant Pests and Diseases, Faculty of Agriculture, Tadulako University Palu, 94118 Central Sulawesi, Indonesia
Flora Pasaru
Department of Plant Pests and Diseases, Faculty of Agriculture, Tadulako University Palu, 94118 Central Sulawesi, Indonesia