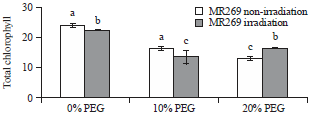
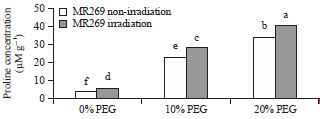
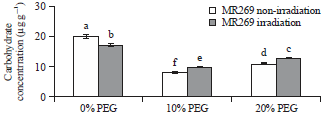
Research Article
Effect of Irradiation and Polyethylene Glycol on Drought Tolerance of MR269 Genotype Rice (Oryza sativa L.)
School of Biosciences and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, Malaysia
LiveDNA: 964.12048
Che Radziah Che Mohd Zain
School of Biosciences and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, Malaysia
Arshad Naji Alhasnawi
School of Biosciences and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, Malaysia
LiveDNA: 60.12099
Anizan Isahak
School of Environmental Science and Natural Resources, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, Malaysia
Mehdi Farshad Ashraf
School of Biosciences and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, Malaysia
Azhar Mohamad
Malaysian Nuclear Agency, Malaysia
Febri Doni
School of Biosciences and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, Malaysia
Wan Mohtar Wan Yusoff
School of Biosciences and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, Malaysia