Research Article
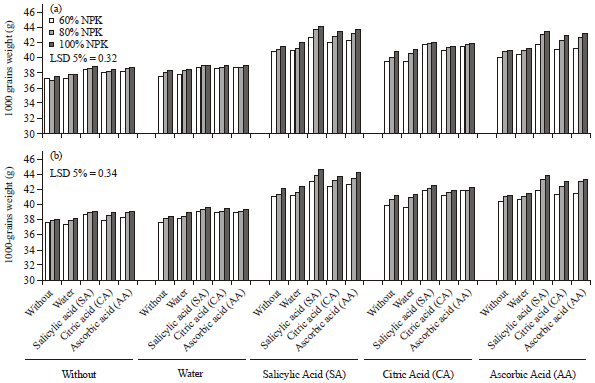
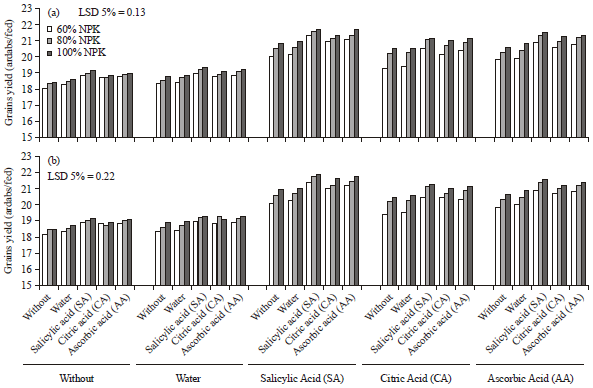
Influence of Antioxidants on Wheat Productivity, Quality and Seed-Borne Fungi Management under NPK Fertilization Levels
Department of Agronomy, Faculty of Agriculture, Mansoura University, Egypt
M. A. El-Metwally
Department of Mycological Research, Plant Pathology Research Institute, Agricultural Research Center, Giza, Egypt