Research Article
Adaptations in Response to Salinity in Safflower Cv. Bhima
Department of Botany, Post Graduate Research Centre, Modern College, Shivajinagar, Pune 411005, M. S. India
INTRODUCTION
Salinity primarily occurs in arid and semiarid regions of the world (Lauchli and Epstein, 1990) and is a part of natural ecosystem (Pathak and Rao, 1998). About 20% of cultivated land is affected by high salt concentration which reduces plant growth limiting crop production (Tanji, 1990; Shannon, 1997; Munns, 2002; Qadir and Murtaza, 2000). According to Niknam and McComb (2000) study of growth and survival under salinity stress condition is necessary as it is necessary to integrate regulation of many physiological mechanisms occurring within the plant. Among the causes of soil salinity are the increasing use of poor quality water, continuous addition of waste salts to our environment and increasing contamination of underground water sources (Somers, 1979). Another reason is the excessive presence of sodium salts like chlorides, sulphates, carbonates and magnesium caused by the capillary rise of salts from underground water into the root zone due to excessive evaporation.
Salinity reduces the ability of plants to utilize water and causes reduction in growth rate and metabolic processes (Munns, 1993, 2002). Plants under saline conditions are stressed due to water deficit, phytotoxicity of Na and Cl ions and nutrient imbalance by limiting uptake and/shoot transport (Munns and Termaat, 1986; Lauchli, 1986; Marschner, 1995). Na competes with K for binding sites essential for cellular function (Tester and Davenport, 2003). According to Gauch and Wadleigh (1944), Magistad (1945), Brown and Hayward (1956) and Bernstein (1961), accumulation of soluble salts in soil increases osmotic pressure of soil solution (Osmotic stress) reducing uptake of water and nutrients by plants. On the other hand, plants absorb constituents of saline solutions at different degrees which brings a toxic or nutritional effect on the plants which is known as specific ion effect (Eaton, 1942; Uvhits, 1946) and nutritional imbalance or a combination of these factors (Ashraf, 1994).
The effect on plant growth and development can be physiological, biochemical (Gorham et al., 1985; Munns, 2002; Munns and James, 2003) and at molecular level (Mansour, 2000; Tester and Davenport, 2003).
Around 50% of the world’s production of safflower is in India. An area of about 0.9 million hectare produces an annual safflower crop fluctuating between 0.35 to 0.50 million tonnes (Malik, 1995). Besides India, USA, Mexico, Ethiopia, Russia and Australia also cultivate safflower. In India the states of Maharashtra, Karnataka and Andhra Pradesh are the dominant safflower producers (74% of the area and 69% of the production).
In India when seed oil is the object, yields are about 90-130 kg florets / hectare and 440 to 660 kg of seed/hectare (CSIR, 1948-1976). In Maharashtra state 310,000 hectares of land is under safflower cultivation from which 47,000 tonnes of seeds are harvested. To overcome the problems of salinity, commercial crop yield for establishing salt tolerance is one of the remedies according to Mass and Hoffman (1977). It is very important to investigate the physiological basis of salt tolerance, in order to study the aspect of salt tolerance which can help to solve salinity problem. One of the useful strategies to combat soil salinity is to select salt tolerant crop/s. The variability in salt tolerance among varieties of crops offers excellent ground for growers to grow salt tolerant cultures to increase agricultural productivity under unfavorable environment like salinity.
The regulation of the transport and distribution of the ions in the various organs of the plants and within the cells is an essential component of the mechanism of salt tolerance. Various aspects of salt tolerance remain uncertain because of the lack of information on the concentration of salt ions in the various organs and compartments. With new dimensions to research technology being available, it is but natural that ‘stress physiology’ studies also incorporate the same but, it is necessary to determine salt tolerance limits and study the interrelations of ion status with other plant metabolism. With this view in mind macro and micro nutrients were analyzed in Carthamus tinctorius var. Bhima. under saline conditions.
MATERIALS AND METHODS
Forty two square cement pots without holes of 30" x 10’= were selected and filled with homogeneously mixed air dried soil and well rotted compost in 3:1 proportion. In each pot, thirty seeds were sown at equal distance and depth and were watered every alternate day on the basis of water holding capacity of soil. After seven days of germination, uniform eight seedlings were maintained in each pot by removing others.
After fifteen days of germination, one set of plants was treated with sodium chloride while other set was treated with sodium sulphate salt. At the same time the control pots were irrigated with equal amount of tap water. The treatments were given to the plants according to the description given in the (US Salinity Laboratory Staff, 1954). The Electrical conductivity of soil in pot was raised from ECe 5.0, 7.5, 10.0, 12.5, 15.0 and 17.5 mS cm-1(USDA Hand book No. 60) by adding required amount of both the salts separately. All the treatments were given in triplicate.
At the time of flowering, these plants were harvested, separated into individual plant parts, sun dried and finally oven dried to constant weight (Sestak et al., 1971). The dried plant parts were subjected to tri acid digestion as per the method given by Chapman and Pratt (1961). Sodium and Potassium were estimated Spectrophotometrically (Model CL:22A, ELICO). Phosphorus was determined by Vanado-molybdate method of Yoshida et al. (1976). Chloride was estimated by the method of Volhard (1956) while sulphate by the method of Chapman and Pratt (1961) and elements like Ca, Mg, Mn, Fe, Ni and Zn by using (Atomic absorption spectro-photometer, Model-300, USA.). All the elements were estimated thrice from the vegetative plant parts like root, stem and fully expanded third leaves. The mean value recorded is the mean of three determinations.
Sodium is a essential microelement. It can partially replace potassium in many reactions known to require potassium. It also plays a role in maintaining a favorable water balance in plants. Increase in sodium content in different plant parts has been reported by Strogonov(1964) and Robinson et al. (1983). According to Abbas et al. (1991) there was continuous increase in Na concentration in all organs of Phaseolus vulgaris with increasing salinity levels in growth medium with more profound increase in roots than in leaves. Reverse was observed when medium was desalinized. According to Jeschke and Pate (1991) Na+ is retained in the root, lateral uptake from xylem by hypocotyl, stem internodes and petioles leading to low intake by young leaf laminae and substantial cycling from older leaves back to the root, at late vegetative growth when Ricinus communis L. plants were exposed to mean salinity stress of 128 moL m-3 NaCl.
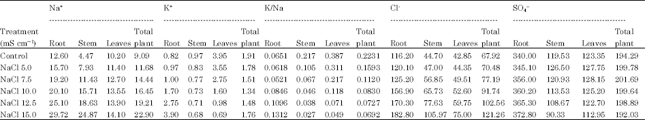
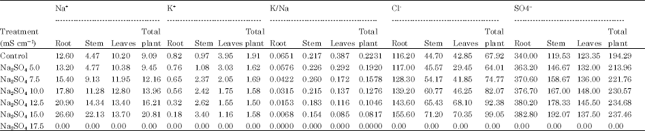
Glenn et al. (1992) observed low Na+ levels in leaf and stem tissues in 3 sub species (Canascens, macropoda, linearis) when grown in 22, 180, 540 and 720 moL m-3 NaCl at harvest concluding the high tolerance of species was not necessarily dependent on high levels of Na+ accumulation. Shitole and Shinde (1991) observed increase in Na in roots under chloride salinity and Na in petiole under sulphate salinity in Carica papaya cv. Ranchi. Yin et al. (1993) observed that Na+ was rapidly absorbed from soil in roots and older leaves with exclusion of K+. Ashraf and Fatima (1995) reported that salt tolerant (260622 and 305167) accessions of Carthamus tinctorius L. accumulated significantly greater Na+ in leaves compared to salt-sensitive (199952 and 170274) accessions. Therefore, salt tolerance of safflower is associated with inclusion of Na+ in leaves. Salt tolerance in glycophytes is associated with the ability to limit uptake and/or transport of saline ions from root zone to shoot (Greenway and Munns, 1980) Results of the present investigation (Table 1-4) also suggested that Na content was more in roots than stem and leaves under both the salinities indicating that roots have capacity to sequester high levels of Na in roots of Carthamus tinctorius var. Bhima. Accumulation of more Na content in roots than stem and leaves suggests tolerance of safflower cv. to both the salinities.
The role of K+ in growth and metabolism makes it difficult to trace a specific and casual relationship between K+ nutrition and the response mechanism. Decrease in K+ content under saline conditions was reported by Robinson et al. (1983) in Spinacea oleraceae. Weimberg (1987) reported that, in Tritium turgidum, K+ concentration decreased with increasing Na+ concentration but their sum remained constant at all salinity levels but in T. aestivum K+ decreased more rapidly than Na+. Abbas et al. (1991) reported decrease in K+ concentration with increasing salinity levels in growth medium except 2nd and 3rd trifoliate leaves in Phaseolus vulgaris. This effect was reversed when medium was desalinised. Abd El-Samad (1993) reported decrease in K+ ion content in Triticum vulgaris L. plants under NaCl salinity. Yin et al. (1993) observed that K+ was excluded with Na+ accumulation. Taleisnik and Grunberg (1994) reported lower K+ uptake rate in Ace and Edlkarin cultivars of Lycospersiion esculentum especially in cv. Ace. at 25 or 100 mM NaCl when compared to control. Reimann and Breckle (1995) reported decrease in potassium content in Salsola kali L. at 200 moL m-3 NaCl. Abd El-Samad and Shaddad (1997) found that sensitivity of soybean cultivar Kint was due to decreased K+ content under NaCl stress. Khan et al. (1997) observed that K+ accumulation decreased in three cultivars of rice subjected to 0 to 200 mM NaCl concentrations. Our results also indicated that, in general, the K+ content in safflower cv.
| Table 1: | A comparative study of the effect of NaCl salinity on Na+, K+, K/Na, Cl- and SO4-- content in Carthamus tinctorius L. cv. Bhima |
 | |
| Root LSD = 0.233, Root LSD = 4.7321, Root LSD = 0.19281, Root LSD = 321.5931, Root SD=777.4355, Stem LSD = 28.028, Stem LSD = 1.599, Stem LSD = 0.2850, Stem LSD = 151.658, Stem LSD = 174.9441, Leaf LSD = 27.57, Leaf LSD = 5.60, Leaf LSD = 2.46, Leaf LSD = 120.10, Leaf LSD = 269.06 | |
| Table 2: | A comparative study of the effect of NaCl salinity on Ca++, PO4--, Zn++, Fe++ and Mn++ content in Carthamus tinctorius L. Cv. Bhima |
 | |
| Root LSD = 83.9605, Root LSD = 8.4532, Root LSD = 4.2769, Root LSD = 4.5420, Root LSD = 0.0993, Stem LSD = 123.3166, Stem LSD = 19.7148, Stem LSD = 6.2004, Stem LSD = 16.4531, Stem LSD = 0.3334, Leaf LSD = 82.22, Leaf LSD = 5.75, Leaf LSD = 3.90, Leaf LSD = 4.75, Leaf LSD = 0.25 | |
| Table 3: | A comparative study of the effect of Na2SO4 salinity on Na+ , K+ , K/Na, Cl- and SO4-- content in Carthamus tinctorius L. Cv. Bhima |
 | |
| Root *LSD = 0 .9423, Root LSD = 1.2987, Root LSD = 0.091, Root LSD = 292.2363, Root LSD = 804.1673, Stem LSD = 27.5927, Stem LSD = 5.2001, Stem LSD = 1.2456, Stem LSD = 127.0146, Stem LSD = 353.2613, Leaf LSD = 26.39, Leaf LSD = 5.35, Leaf LSD = 0.49, Leaf LSD = 113.10, Leaf LSD = 299.15 | |
| Table 4: | A comparative study of the effect of Na2SO4 salinity on Ca++, PO4--, Zn++, Fe++ and Mn++ content in Carthamus tinctorius L. Cv. Bhima |
 | |
| Root LSD = 94.4595, Root LSD = 11.3902, Root LSD = 5.7513, Root LSD = 6.2703, Root LSD = 0.0993, Stem LSD = 181.9044, Stem LSD = 28.7355, Stem LSD = 5.8646 Stem LSD = 15.1213, Stem LSD = 0.3537, Leaf LSD = 90.86, Leaf LSD = 6.78, Leaf LSD = 4.75, Leaf LSD = 4.00, Leaf LSD = 0.31 | |
Bhima was less than the control at all levels of NaCl and Na2SO4 reflecting that K+ uptake fails under saline conditions.
According to Strogonov (1964) and Brun (1987) K+ increases under saline conditions in salt tolerant plants. According to Robinson et al. (1983) K+ was the predominant monovalent cation at 160-200 mM in Spinacea oleraceae, Beta vulgaris and Pisum sativum. Jeschke and Wolf (1988) observed that when Ricinus communis L. was grown at 160 moL m-3 NaCl, K+ concentration increased in leaf blades. K+ concentration in both long distant transport fluids were maintained at high levels. Shitole and Shinde (1991) observed that cv. Ranchi of papaya has efficient K+ uptake mechanism under saline conditions. Glenn et al. (1992) reported high K+ levels and low Na: K ratios in leaf and stem tissues in Canascens, macropoda, linearis subsp of Atriplex canascens when grown in 22, 180, 150 and 720 moL m-3 NaCl at harvest. Storey et al. (1993) reported that shoot K+ concentration was maintained over a range of salinity’s upto 400 moL m-3 followed by increase in external osmotic pressure in Melanthera biflora Asteraceae. Baset and Arju (1990) have reported low K:Na ratio in mid portion of 3rd youngest leaf in two rice varieties grown in nutrient solution without and with 50 mM NaCl. Zhang et al. (1996) observed enhanced Na:K ratio in Eleucine coracana L. under NaCl stress. According to Jeschke et al. (1986) K:Na ratio was higher in leaflets than in adjoining petiole and stem segments and in younger than in older parts of the shoot suggesting capacity of Na+ retention in stem and selectivity in K+ mobilization to young tissues with increasing salinity (NaCl 1,5,10,25,40 moL m-3 ) in Lupinus albus cultivar Ultra. From these references it is clear that in many plants K: Na ratio increases under saline conditions. Generally these plants are salt susceptible and lack selective absorption mechanisms. Results of the present investigation on Carthamus tinctorius var. Bhima (Table 1, 3) revealed that average K:Na ratio of total plant decreased with increase in salt concentrations of both the salts which indicated that there is no selective absorption mechanism in Carthamus tinctorius cv. Bhima. However the plant has the ability to maintain more growth with less K:Na ratio upto ECe 7.5 m Scm-1 of Na2SO4. This is due to its genetic potential. Decreased K:Na ratio reduced productivity at all levels of NaCl salinity (Table 5). This indicates that this variety is susceptible to NaCl salinity.
Chloride is not very essential micronutrient in higher plants but it is known to perform some important functions (Abbas et al., 1991) observed continuous increase in Cl-- concentration in all organs of Phaseolus vulgaris with increasing salinity levels in growth medium. (Gouia et al., 1994) observed accumulation of Cl-- in all parts of bean plants at 50 mM NaCl. According to Hu and Schmidhalter (1988) Cl-- concentration (m mol kg-1 fresh weight) was high at 120 mM than at 0 mM NaCl along leaf axis from leaf base of wheat and local net deposition rates of Cl-- ( m mol kg-1 fresh weight h-1) in actively elongating zone were enhanced by 120 mM NaCl. Higher Cl-- tissue concentration did not result in ion toxicity in growing leaves but could have caused ion imbalance. Ashraf and Fatima (1995) found that salt tolerant (260622 and 305167) and salt sensitive (199952 and 170274) accessions of Carthamus tinctorius L. did not differ in Cl-- concentrations. Results of present investigation show that chloride uptake was stimulated at all levels of chloride and sulphate salinizations in Carthamus tinctorius cv. Bhima. However, it was more accumulated in roots which reflected that salt tolerance mechanism of this variety is associated with exclusion of Cl from leaves.
Gauch and Wadleigh (1944) reported that sulphate is absorbed in much smaller quantities than chloride but produces equivalent effects on growth of bean plants. In 1945, they observed exponential increase in sulphate content in leaves and roots with increasing concentration of sulphate in solution whereas the stem showed linear increase. According to Hayward and Wadleigh (1949) sulphate ion restricts the absorption of Ca++ while it promotes uptake of Na+. At the same time (Richards, 1954) reported limited uptake of Ca++ under sulphate salinity which is responsible for the sensitivity of the plants because stimulated uptake of Na+ disturbs cationic balance in salinized plant tissues. Sulphate contents in bean (Meiri et al., 1971) and in Cucumis melo (Nukaya et al., 1984) was increased with increasing sulphate salinization.
Our results suggested that all concentrations of sodium chloride and sulphate stimulated sulphate uptake. (Table 1-4) which reflects that plants have ability to maintain sulphate uptake under saline conditions.
Ca++ is important to plants growing under saline environment (Rains, 1972). Exogenous Ca++ reduces the perception of stress by the cytoplasm. It has been suggested that Ca++ displaces Na+ from the plasmalemma of salt stressed root cell, thus decreasing the influx of ions into the cytoplasm (Lynch et al., 1987). Extra Ca++ added to the medium possibly has some role in maintaining membrane integrity which contributes to the ability of different plants to resist salt stress. The presence of extra Ca++ in the solution lowers response to osmotic stress.
Abbas et al. (1991) reported decrease in Ca++ concentration with increasing salinity levels except in 2nd and 3rd trifoliate Phaseolus vulgaris leaves. This effect was reversed when medium was desalinized. Gouia et al. (1994) observed decrease in Ca++ cations in bean plants at 50 mM NaCl. Lopez and Satti (1996) observed reduction in calcium concentration when 5 cultivars of tomato were subjected to 50 mM NaCl.
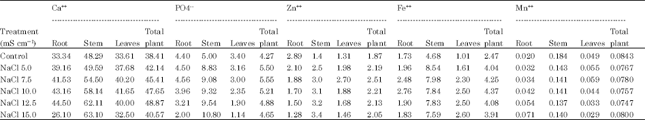
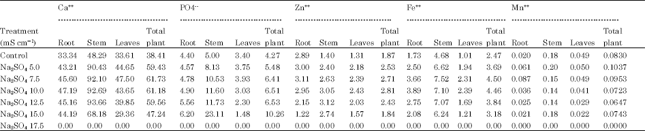
Shitole and Shinde (1991) observed that salt stress caused accumulation of calcium in all parts of papaya plant. According to Hu and Schmidhalter (1988) Ca++ concentration (m mol kg-1.fresh weight) was high at 120 mM than at 0 mM NaCl along leaf axis from leaf base of wheat and local net deposition rates of Cl¯G (m mol Kg-1 fresh weight h-1) in actively elongating zone were enhanced by 120 mM NaCl. However, high concentration of Cl-- in tissue cause ion imbalance but did not result in ion toxicity in growing leaves. Hamada and El-Enany (1994) reported that calcium content remained unaffected by salinity in pea plants. Ashraf and Fatima (1995) reported no difference in calcium concentration in salt tolerance and salt-sensitive safflower at 0, 70, 140 and 120 mol m-3 NaCl. Results of the present investigation (Table 1-4) revealed that at all levels of both the salinizations, Ca uptake was more. Thus, plants can survive upto ECe 10.0 mS cm-1 of NaCl and upto ECe 12.5 mS cm-1 of Na2SO4 by maintaining more Ca uptake under saline conditions.
Reduction in P content under saline conditions was reported by Strogonov (1964) and Shitole and Shinde (1991), Ferguson and Hedlin (1963) have studied the influence of NaCl and Na2SO4 salts on P absorption in barley and found that P absorption was favored at lower concentration (upto 6 mm hos cm) of the salts while it was adversely affected at high concentrations. Wilson et al. (1970) has suggested that higher P content was closely related to salt tolerance of Glycine falcata. Shimose (1972) observed no significant effect of Na2SO4 treatment on P contents of barley, wheat and asparagus. Similar observations were made by Matar et al. (1975), Abdel Rehman (1987), found no effect of P in leaves of cowpea under NaCl salinity.
However, the P content (Table 1-4) was more than the control at all levels of NaCl and Na2SO4 indicating that P uptake is stimulated in safflower cv. Bhima which is one of the reason for increased productivity upto ECe 7.5 mS cm-1 of Na2SO4.
Early reports indicate both primitive and adverse effects of Zinc uptake in plants under saline conditions. A positive correlation between soil salinity and zinc was reported by Hassan et al. (1970) in the leaves and stems of corn and barley. Cramer et al. (1991) observed increase in Zn over time in Hordeum vulgare L. (M72) for 25 days treated with NaCl or KCl (125 mM). According to Venkatesan et al. (1997) Zinc content upto optimum salt level in Ipomoea pes-caprae sweet plants. Gadallah and Ramadan (1997) reported that high concentration of Zinc in Carthamus tinctorius L. improved growth of roots and enhanced xylem formation in NaCl stressed plants and compared with plants grown without Zinc.
Results of the present (Table 1-4) investigation indicated that under NaCl treatment, in roots, Zn is decreased whereas, in stem and in leaves Zn content increases with increasing salinity. Under sulphate salinity, however, in roots low levels stimulated while high levels decreased Zn content. In stem and leaves Zn content increased with increasing levels of salinity in Carthamus tinctorius cv. Bhima.
Shimose (1972) reported decrease in Fe++ content under saline conditions. Medium levels of salinity resulted in highest content of Fe++ while higher levels of salinity, decreased Fe++ content of shoots of Sankha 8, Sonalika, Sakha 3 and Soltane cultivars of wheat (El-Sherbiery et al., 1986). On the contrary, increased iron content have been observed by Mass et al. (1972) in the roots and tops of tomato and soybean. Cramer et al. (1991) observed increase in Fe++ in Hordeum vulgare L. (M72) grown for 29 days and treated with NaCl or KCl (125 mM). According to Venkatesan et al. (1997) increase in Fe++ content was reported upto optimum salt level in Ipomoea pes-caprae sweet plants.
Results of the present investigation (Table 2, 4) revealed that Fe++ uptake is stimulated at all levels of both the salts in Carthamus tinctorius L. Var. Bhima. This stimulation of Fe++ uptake may be due to abrupt changes in membrane permeability under saline conditions. Results also indicated that Fe++ is not much stored in roots but it is translocated to stem. Within the stem Fe++ is more retained under both the salinizations indicating the presence of some regulatory mechanism within the stem.
Inhibition of Mn++ uptake under saline conditions was reported by Shimose (1972) in barley under saline conditions. Cramer et al. (1991) grew Hordeum vulgare L. (M72) for 25 days and treated with NaCl or KCl (125 mM) and reported decline in Mn concentration over time in shoot below 50 mmol g-1 fresh weight.
Stimulation of Mn++ uptake under saline conditions was reported by Mass et al. (1972) in tomato and soybean. Venkatesan et al. (1997) reported increase in Mn content upto optimum salt level in Ipomoea pes-caprae sweet plants. Niazi and Ahmed (1985.) reported that Mn++ uptake in stem, leaves and fruits of tomato under NaCl 90, 5, 10, 25, 50 meq L-1) remained unaffected.
Results of present investigation (Table 1-4) indicated Mn++ uptake was more than the control in plants grown upto ECe 7.5 mS cm-1 of sulphate and was less than the control at all high levels. Thus increased Mn++ uptake at low levels of sulphate helps for increase in productivity (Table 5, 6). One of the reasons for decreased productivity at all levels of NaCl and higher levels of Na2SO4 must be due to decreased Mn uptake.
From the above results it is clear that the average Na and Cl content (Table 1, 3) of the total plant increased with increasing levels of chloride and sulphate salinity thereby indicating safflower cv. Bhima does not have selective absorption mechanisms under saline conditions. However, it adapts to saline conditions by storing more Na and Cl in roots and stem under both the salinizations.
Accumulation of ions in roots in excessive amounts caused decrease in dry weight reducing productivity at all levels of NaCl and at higher concentration of Na2SO4 (above ECe 10.0 mS cm-1) (Table 5, 6).
| Table 5: | Effect of NaCl salinity on productivity of stem, root, leaf and total plant in Carthamus tinctorius Var. Bhima at flowering |
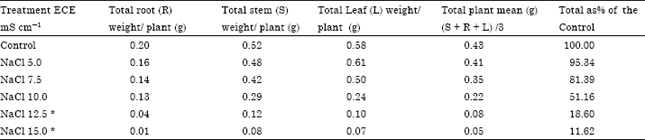 | |
| Plants died during 60 to 90 days of growth. Root LSD = 0.029 Stem LSD = 0.027 Leaf LSD = 0. 020 | |
| Table 6: | Effect of Na2SO4 salinity on productivity of stem, root, leaf and total plant in Carthamus tinctorius Var. Bhima at flowering |
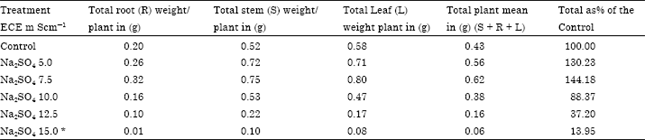 | |
| Plants died during 60 to 90 days of growth., Root LSD = 0.025 Stem LSD= 0.021 Leaf LSD= 0. 025, *LSD Least significant difference | |
Sulphate and calcium content (Table 1-4) was linearly increased with increasing concentrations of both the salts thereby indicating that all levels of sulphate and chloride stimulate sulphate uptake in safflower cv. Bhima. The average content of the total plant was more than the control at all levels of chloride and sulphate salinity indicating that plants adapt to saline conditions by maintaining efficient sulphate and calcium uptake. Translocation of phosphorus from roots and stem to leaves reduced under both the salinizations which must be one of the reasons for decreased productivity at all higher levels of both the salts (Table 2, 4).
All levels of both the salts stimulate uptake of zinc and iron in Carthamus tinctorius Var. Bhima. However Mn uptake was different under chloride and sulphate salinizations (Table 2, 4).
From the above results it is clear that the average Na and Cl content (Table 1, 3) of the total plant increased with increasing levels of chloride and sulphate salinity thereby indicating safflower cv. Bhima does not have selective absorption mechanisms under saline conditions. However, it adapts to saline conditions by storing more Na and Cl in roots and stem under both the salinizations. Accumulation of ions in roots in excessive amounts caused decrease in dry weight reducing productivity at a ll levels of NaCl and at higher concentration of Na2SO4 (above ECe 10.0 m Scm-1) (Table 5, 6).
Sulphate and calcium content (Table 1-4) was linearly increased with increasing concentrations of both the salts thereby indicating that all levels of sulphate and chloride stimulate sulphate uptake in safflower cv. Bhima. The average content of the total plant was more than the control at all levels of chloride and sulphate salinity indicating that plants adapt to saline conditions by maintaining efficient sulphate and calcium uptake. Translocation of phosphorus from roots and stem to leaves reduced under both the salinizations which must be one of the reasons for decreased productivity at all higher levels of both the salts (Table 2, 4).
All levels of both the salts stimulate uptake of zinc and iron in Carthamus tinctorius var. Bhima. However Mn uptake was different under chloride and sulphate salinizations (Table 2, 4).