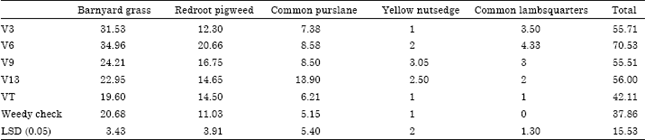
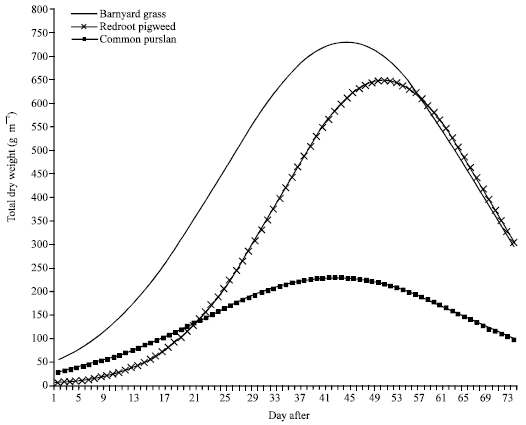
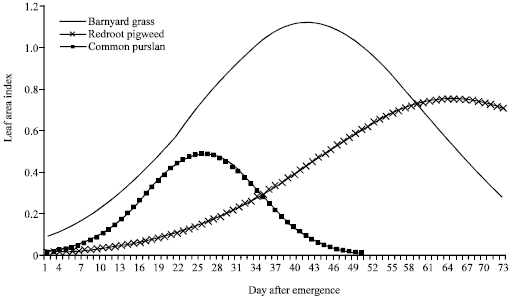
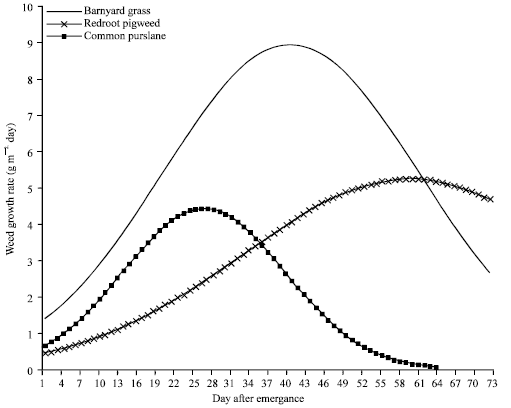
Research Article
Evaluating Weeds Competitive Ability in a Corn Field in Southern West of Iran
Department of Weed Identification and Control, Faculty of Agriculture, Azad Islamic University of Shushtar, Iran
S. Lorzadeh
Department of Weed Identification and Control, Faculty of Agriculture, Azad Islamic University of Shushtar, Iran
N. Aryannia
Department of Weed Identification and Control, Faculty of Agriculture, Azad Islamic University of Shushtar, Iran