Research Article
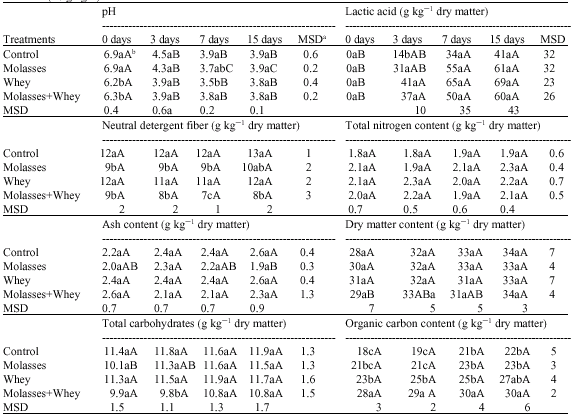
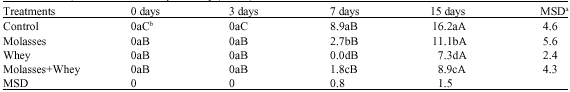
Effect of Sugarcane Molasses and Whey on Silage Quality of Maize
Instituto Tecnologico de Tuxtla Gutierrez, Carretera Panamericana Km. 1080, Tuxtla Gutierrez, Chiapas CP 29050, A.P. 599, Mexico
Mario A. Cobos
Colegio De Posgraduados, Estado de Mexico, Mexico
Lucia Maria Cristina Ventura-Canseco
Instituto Tecnologico de Tuxtla Gutierrez, Carretera Panamericana Km. 1080, Tuxtla Gutierrez, Chiapas CP 29050, A.P. 599, Mexico
Teresa Ayora-Talavera
Instituto Tecnologico de Tuxtla Gutierrez, Carretera Panamericana Km. 1080, Tuxtla Gutierrez, Chiapas CP 29050, A.P. 599, Mexico
Miguel Abud-Archila
Instituto Tecnologico de Tuxtla Gutierrez, Carretera Panamericana Km. 1080, Tuxtla Gutierrez, Chiapas CP 29050, A.P. 599, Mexico
Maria Angela Oliva-Llaven
Facultad de Medicina Veterinaria y Zootecnia, Universidad Autonoma de Chiapas, Chiapas, Mexico
Luc Dendooven
Departamento de Biotecnologia y Bioingenieria, CINVESTAV, Mexico DF, Mexico
Federico A Gutierrez-Miceli
Instituto Tecnologico de Tuxtla Gutierrez, Carretera Panamericana Km. 1080, Tuxtla Gutierrez, Chiapas CP 29050, A.P. 599, Mexico









khezer molaei Reply
The first time you've come to the site. I think a good site is filled with content. thank you from the efforts. Bye