Research Article
Comparative Study on the Harmful Effect of Salt Ingestion and its Dietary Restriction in Wistar Rats
Department of Medical Biochemistry, College of Medicine, Ambrose Alli University, Ekpoma, Edo, Nigeria
The relationship between mean sodium intake and mean blood pressure has been assessed and a positive correlation was the finding across a broad range of populations and among economically developed and underdeveloped groups of communities (Law et al., 1991). The dispute that has lasted over century on the evidence that suggests that hypertension is in part due to the present high intake of salt has been resolved through scientific studies. In fact, there is a consensus that dietary sodium (as in salt) plays a significant role and is responsible for the much of the rise in blood pressure (De Wardener and MacGregor, 2002; Tuomilehto et al., 2001). A large proportion of patients with primary hypertension are characterized by insulin resistance (Shen et al., 1988; Ferrannini et al., 1987) which is suggested to be the cause of a cluster of cardiovascular risk factors called “Metabolic syndrome”. In addition to disturbances in glucose metabolism and hypertension, this syndrome includes dyslipidemia, abdominal obesity and microalbuminuria (Alberti and Zimmet, 1998).
Against this background, this study was therefore undertaken to investigate the effect of salt inclusion in the diet and it restriction (reversal effect) on body weight, pulse rate, blood glucose and lipid profile using Wister rats as a model. Significantly, the result may be very important for dietary guidelines for persons at risk for the metabolic syndrome and cardiovascular disease indicated by body weight, blood glucose, pulse rate and lipid profile.
Experimental animals: Adult Wistar rats (N = 18) of comparable weight (175±50) were procured from animal unit of the Department of Anatomy, University of Benin, Benin City, Nigeria. They were moved to the research laboratory of Anthonio Research Centre, Ekpoma, Nigeria, where they were allowed two weeks of acclimatization and fed with feed and water ad libitum.
They were housed under standard environmental conditions in a well ventilated room under a 12/12 h light/dark cycle.
Animal grouping and experimental procedure: During the acclimatization period, Wistar rats were divided into three groups of 6 rats each and allowed to complete the two weeks of acclimatization.
Group A (n = 6) served as the control and received control diet (100 g feed) with water given ad libitum for six weeks.
Group B (n = 6) received test diet (92 g feed+8 g salt) with water given ad libitum for six weeks.
Group C (reversal group) received test diet (92 g feed+8 g salt) for the 1st three weeks and then was on control diet (100 g feed) for the next three weeks with water given ad libitum.
Sample collection and analysis: Prior to the study (base value), three week into the study (mid value) and at the end of the study (final value), body weight, pulse rate and blood glucose were determined using electronic weighing scale, palpation method and gluco-meter, respectively following standard procedures.
Lipid profile was determined at the end of the study.
Statistical analysis: Data was analyzed using the Statistical Package for Social Science (SPSS) version 17 for the analysis. The one way analysis of variance was used and the level of significance was placed at p less than 0.05.
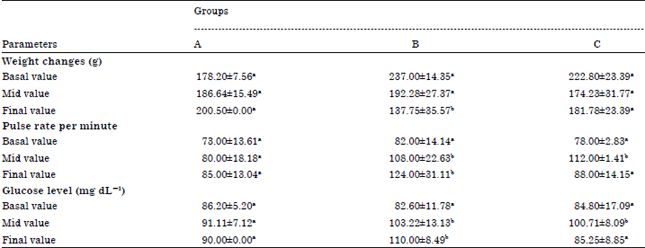
Table 1 shows the effect of salt ingestion and salt restriction on weight of adult Wistar rats compared with control. A steady mean body weight gain was observed in the control. Group B presented a steady mean body weight loss from 237.00±14.35 g before salt ingestion to 192.28±27.37 g during the 1st three weeks of salt ingestion to a significant weight loss of 137.75±35.57 g at the end of the sixth week. However, group C presented a non significant weight loss (174.23±31.77 g) during the time of salt ingestion and a non significant weight again (181.78±23.39 g) during the reversal effect.
Table 1 shows the effect of salt ingestion and it restriction on pulse rate of adult Wistar rats compared with control. A non significant increase in pulse rate was observed in the control but the ingestion of salt (group B) brings about a significant increase (p<0.05) in pulse rate. In group C, three weeks of salt ingestion resulted in significant increase in pulse rate (112.00±1.41 min-1) while the withdrawal of salt from the diet brought about a reduction (88.00±14.15 min-1) compare to when salt was ingested (112.00±1.41 min-1).
Table 1 shows the effect of salt ingestion and its restriction on blood glucose of adult Wistar rats compared with control. A significant increase (p<0.05) in blood glucose was observed with the ingestion of salt for three weeks; as shown in mid value in groups B (103.22±13.13 mg dL-1) and C (100.71±8.09 mg dL-1).
| Table 1: | Comparative table on weight changes, pulse rate and blood glucose changes in Wistar rats fed salt diet and reversal effect |
 | |
| Values are Mean±SD, values in each group having different superscript are significantly different at p<0.05 | |
| Table 2: | Changes in lipid profile parameters in wistar rats fed salt diet and reversal effect |
 | |
| Values are Mean±SD, values in each group having different superscript are significantly different at p<0.05 | |
The significant increase was potentiated further with continuous ingestion of salt for the next three weeks (group B, 110.00±8.49 mg dL-1) but become similar with the basal value (84.80±17.09 mg dL-1) with the withdrawal of salt from the diet (group C, 85.25±8.85 mg dL-1).
Table 2 shows the effect of salt ingestion and it restriction on lipid profile of adult Wistar rats. On cholesterol, the ingestion of salt for six (group B, 149.36±65.73) and three weeks ingestion plus three weeks withdrawal (group C, 142.54±28.41) brought about a non significant increase in cholesterol compared to the control (127.38±82.82). On triglyceride however, the ingestion of salt for six weeks (group B, 184.69±115.35) resulted in a non significant reduction while a non significant increase was observed with three weeks ingestion plus three weeks withdrawal (group C, 288.42±56.56) compared with the control (225.47±66.96). The ingestion of salt for six weeks (as in group B) has a reducing effect on bad cholesterol (low density lipoprotein) and an increasing effect on good cholesterol (high density lipoprotein). On the other hand, withdrawal of salt ingestion (as in group C) resulted in increase in bad cholesterol (low density lipoprotein) but a reduction in good cholesterol (high density lipoprotein). In another words, salt ingestions has a favourable effect while it withdrawer has a damaging effects on good and bad cholesterols compared to the observation in the control group.
Gain in weight associated with salt and water retention that accompanies cardiac failure has been noted (De Wardener and MacGregor, 2002). In fact, dietary salt ingestion has been reported to induce mean body weight gain (Akpamu et al., 2011) specifically in women (Melander et al., 2000) probably through its involvement in increasing the size of muscle mass (due to the hypertrophy and deposition of collagen and fibrous tissue) and thickening of the coronary arteries (De Wardener and MacGregor, 2002; Houghton et al., 1990; Marcus et al., 1982). However, the finding of this study on the effect of salt on body weight of Wistar rats contradicts this weight inducing facts by previous studies. On the other hand, the present findings on the effect of salt on body weight is in line considering the facts that dietary sodium restriction has been linked with an increased white adipose tissue mass in rats (Prada et al., 2000,2005; Okamoto et al., 2004).
Dietary salt ingestion has been linked with increased in blood pressure and this was the observation of the present study, considering the effect of dietary salt ingestion on pulse rate (Table 1, group B). This finding is in line with several other studies (National Heart Foundation of Australia, 2006; De Wardener and MacGregor, 2002; Tuomilehto et al., 2001). To this regard, many systematic reviews and meta-analyses have been conducted to assess the efficacy of salt reduction on hypertension (He and MacGregor, 2002; Hooper et al., 2002) and advice on dietary salt restriction is effective in lowering blood pressure (Dickinson et al., 2006; Hooper et al., 2004). By implication, dietary sodium restriction was advocated as an important non-pharmacological approach for the prevention and treatment of hypertension (Whelton et al., 1998; Sacks et al., 2001). This advocacy of dietary sodium restriction collaborates with the present finding (Table 1, group C) on the effect of dietary salt restriction on pulse rate in this study.
Earlier studies have suggested that salt sensitivity and insulin sensitivity may be related (Melander et al., 2000). In the present study, the ingestion of salt resulted in a significant (p>0.05) increase in blood glucose while its withdrawal resulted in a reduction in blood glucose level (Table 1). In accordance with the finding of this study, studies have associated primary hypertension with insulin resistance (Shen et al., 1988; Ferrannini et al., 1987) which is suggested to be the cause of a cluster of cardiovascular risk factors called “Metabolic syndrome” (Melander et al., 2000). In addition to the disturbances in glucose metabolism and hypertension cause by salt ingestion, dyslipidemia, abdominal obesity and microalbuminuria have been included (Alberti and Zimmet, 1998).
While study has been unable to detect any unfavourable effects on serum lipids or atherogenic index on salt restriction effects on lipids (Meland et al., 1997), increased levels of total cholesterols and LDL cholesterols are reported in the Cochrane review (Jurgens and Graudal, 2004). In addition, studies evaluating long-term effects of salt moderation are unable to detect significant dyslipidaemic effects (Jurgens and Graudal, 2004; McCarron et al., 1997). In contrast to the results of previous study where hypertension induced by salt ingestion produces dyslipidemia (Alberti and Zimmet, 1998) we showed that salt ingestions has a favourable effect while it withdrawer has a damaging effects on good and bad cholesterols compared to the observation in the control group (Table 2). This suggests that salt inclusion in the diet may have beneficial effects on good and bad lipid cholesterol while salt restriction may have unfavorable effects on good and bad lipid cholesterol.
Conclusively, salt ingestion may be unfavourable on body weight, blood pressure and glucose level but may have a favorable impact on lipid profile. On the other hand, dietary restriction of salt intake may favour body weight, blood pressure and glucose but unfavourable to lipid profile.