Research Article
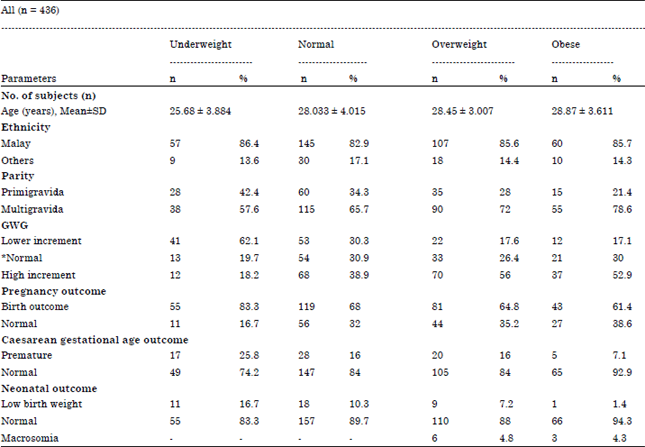
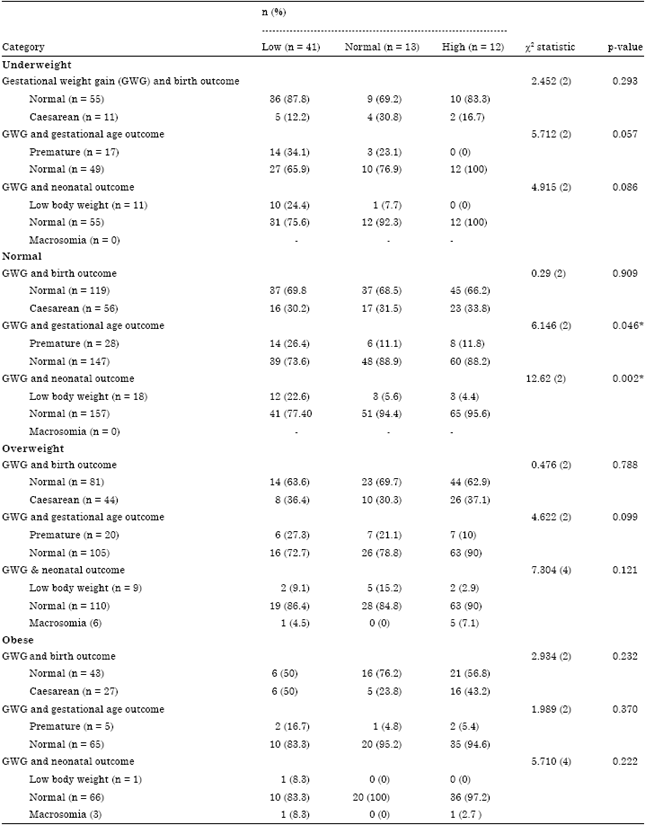
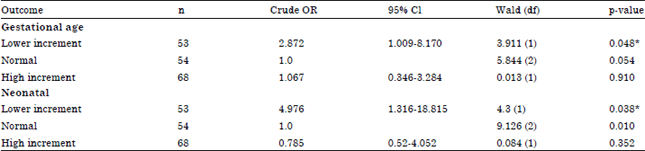
The Association of Gestational Weight Gain and the Effect on Pregnancy Outcome Defined by BMI Group among Women Delivered in Hospital Kuala Lumpur (HKL), Malaysia: A Retrospective Study
Department of Nutrition and Dietetics, Faculty of Health Sciences, Universiti Teknologi MARA, 42300 Puncak Alam, Selangor, Malaysia
Hayati Adilin Mohd Abd Majid
Department of Nutrition and Dietetics, Faculty of Health Sciences, Universiti Teknologi MARA, 42300 Puncak Alam, Selangor, Malaysia
Siti Shafura Abas
Department of Nutrition and Dietetics, Faculty of Health Sciences, Universiti Teknologi MARA, 42300 Puncak Alam, Selangor, Malaysia
Ajau Danis
Department of Basic Sciences, Faculty of Health Sciences, Universiti Teknologi MARA, 42300 Puncak Alam, Selangor, Malaysia
Khairil Anuar Md. Isa
Department of Basic Sciences, Faculty of Health Sciences, Universiti Teknologi MARA, 42300 Puncak Alam, Selangor, Malaysia