Research Article
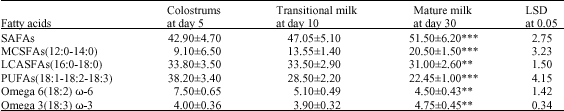
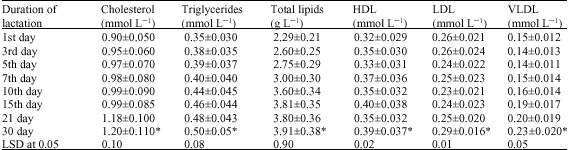
Lipid Fractions and Fatty Acid Composition of Colostrums, Transitional and Mature She-Camel Milk During the First Month of Lactation
Department of Biochemistry, Animal Health Research Institute, Dokki, Giza, Egypt
Omar A. Salama
Animal Production Research Institute, Dokki, Giza, Egypt