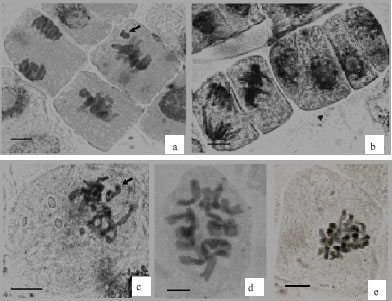
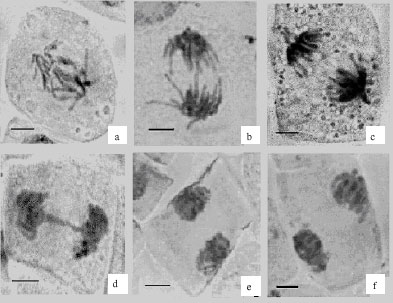
Research Article
Mitotic Changes in Root Meristems of Lens culinaris Treated with Fusilade (fluazifop-p-butyl)
Department of Biology, Faculty of Science and Arts, Kocaeli University, 41380, Umuttepe, Izmit, Kocaeli, Turkey
Nuran Ekici
Department of Science Education, Faculty of Education, Trakya University, 22030, Aysekadin Yerleskesi, Edirne, Turkey
Feruzan Dane
Department of Biology, Faculty of Science and Arts, Trakya University, 22030, Edirne, Turkey