Research Article
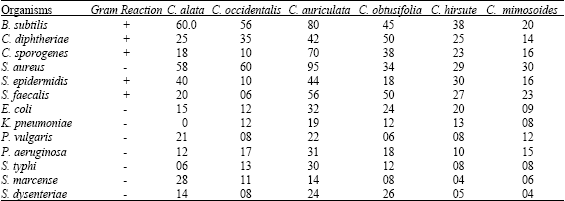
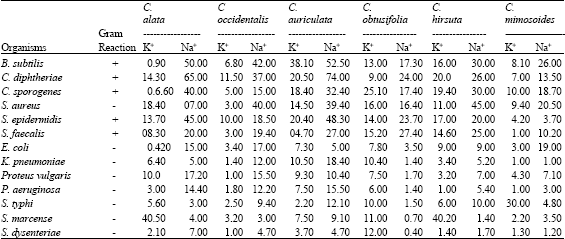
Comparative Studies on the Amount of Protein, Sodium and Potassium Ions Released by Methanolic Extracts from Six Cassia Species
Departmentment of Microbiology, School of Sciences, Federal University of Technology, P.M.B. 704, Akure, Nigeria
F.A. Akinyosoye
Departmentment of Microbiology, School of Sciences, Federal University of Technology, P.M.B. 704, Akure, Nigeria
F.C. Adetuyi
Departmentment of Microbiology, School of Sciences, Federal University of Technology, P.M.B. 704, Akure, Nigeria