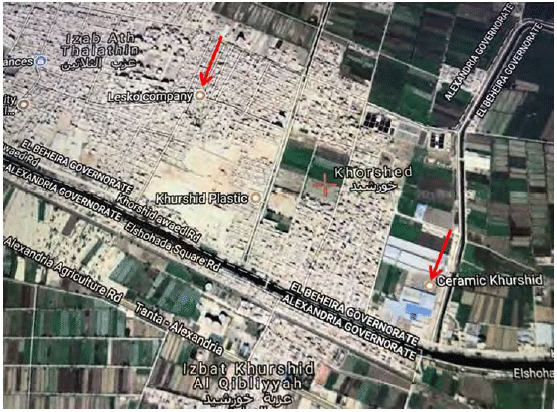
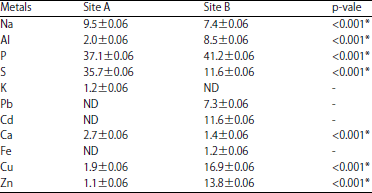
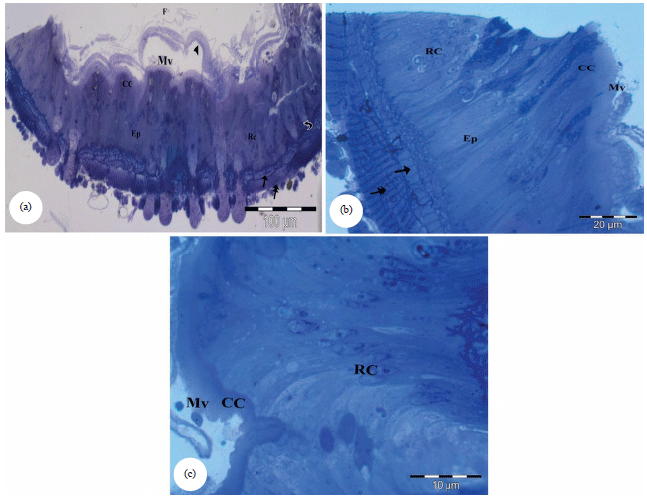
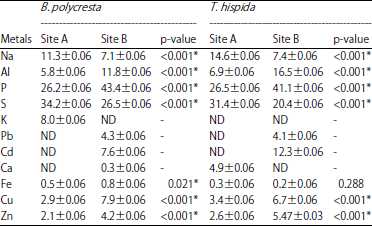
Research Article
Histological and Ultrastructure Alterations in the Midgut of Blaps polycresta and Trachyderma hispida (Coleoptera: Tenebrionidae) Induced by Heavy Metals Pollution
Department of Zoology, Faculty of Science, Alexandria University, 21511 Alexandria, Egypt
LiveDNA: 20.26622
Lamia Mostafa El- Samad
Department of Zoology, Faculty of Science, Alexandria University, 21511 Alexandria, Egypt