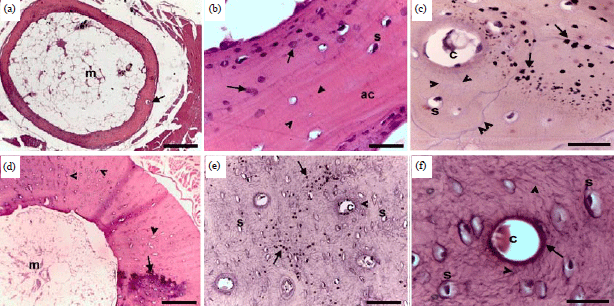
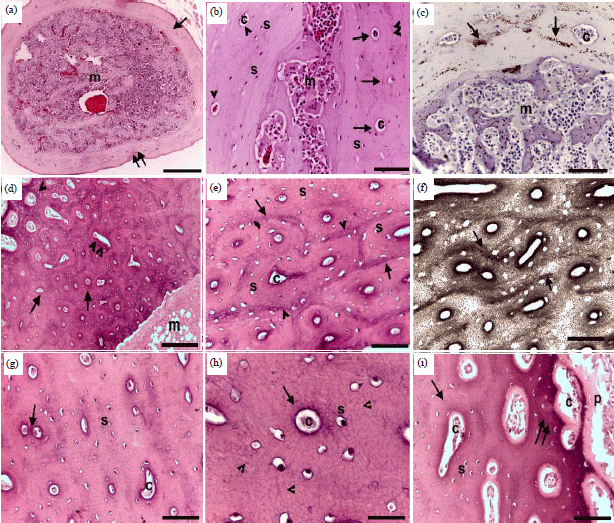
Research Article
Absence of Typical Haversian System from the Compact Bone of Some Reptile and Bird Species
Department of Histology, Faculty of Veterinary Medicine, South Valley University, Qena, Egypt
LiveDNA: 20.1825
Mohamed Abdelsabourv-Khalaf
Department of Anatomy and Embryology, Faculty of Veterinary Medicine, South Valley University, Qena, Egypt
Fatma Khalil
Department of Histology, Faculty of Veterinary Medicine, South Valley University, Qena, Egypt