Research Article
Study of Bacteriocin Produced by Enterococcus faecium Strain Isolated from Traditional Fermented Tomatoes in Algeria
Laboratory of Applied Microbiology, Es-Senia University, Oran, Algeria
LiveDNA: 213.20257
Scientific studies and research revealed the danger of increasing the rate of consumption of preservatives on humans and confirmed that they cause many diseases in the long term. Biopreservation of food is a conservation technology consisting in adding microorganisms selected in foods generally packaged under vacuum or in a protective atmosphere, selected for their ability to inhibit the growth of undesirable microorganisms1-2. The objective is to extend the shelf life of the products and/or to control the development of a specific bacterial hazard3-4. The microorganisms selected for these applications generally belong to the group of lactic acid bacteria, as they are often the majority on packaged refrigerated products and possess inhibition properties5-6. The strains of lactic acid bacteria of interest were selected for their activity of inhibiting target strains belonging to pathogenic and/or spoilage flora, as well as other criteria, such as growth at low temperature or lack of character altering. Numerous studies are devoted to the application of lactic acid bacteria and/or their bacteriocins in the various foodstuffs7-8. Bacteriocins produced by lactic acid bacteria (LAB) have received particular attention in recent years due to their potential application in food industry as natural preservatives. Bacteriocin production could be considered as an advantage for food and feed producers since, in sufficient amounts, these peptides can kill or inhibit pathogenic bacteria that compete for the same ecological niche or nutrient pool. This role was supported by the fact that many bacteriocins have a narrow host range and was likely to be most effective against related bacteria with nutritive demands for the same scarce resources3.
Enterococci are an ancient genus of lactic acid bacteria (LAB) that are highly adapted to living in complex environments and surviving adverse conditions. They are ubiquitous, inhabiting the gastrointestinal tracts of a wide variety of animals, from insects to man9-10. The genus Enterococcus includes a wide range of strains, in some cases suitable as starter cultures or probiotics and in other cases known as spoilage or pathogenic microorganisms. Some enterococci play a positive role in various fermented foods, including dairy and vegetable products, apparently through proteolysis, lipolysis, exopolysaccharide production and citrate breakdown11-12.
Like other lactic acid bacteria, enterococci are known to secrete antimicrobial substances, making them potentially useful for the prevention of bacterial foodborne illness. Many enterococci produce at least one bacteriocin, which is ribosomally synthesized antimicrobial peptide, active against a wide range of foodborne pathogens including Listeria spp.13-14. Most bacteriocins produced by enterococci especially by the Enterococcus faecium strains are grouped as enterocins including bacteriocin T8, enterocin AS-48, enterocin B, enterocin 1071A, enterocin L50, enterocin K1 and enterocin MR1015-18.
Interest in bacteriocin study produced by enterococci has been stimulated by the fact that they can inhibit Gram-positive food-borne pathogens but they are also active against decarboxylase-positive enterococci. In addition, most of enterocins inhibit Gram-positive bacteria. Because of bacteriocins activity against food-borne pathogens on one side and consumer demands for more "natural" preservatives on the other side, bacteriocins have been suggested for use as "biopreservatives"19.
Therefore, the first objective of this study was to determine the inhibitory effect of bacteriocin produced by E. feacium towards micro-organisms in foods environment. The second objective was to characterize the bacteriocin, use some agro-industrial wastes as media in the place of MRS broth to promote bacteriocin production and activity and finally application of this bacteriocin in biopreservation of foods in the future.
Isolation and identification of bacterial strain: Enterococcus faecium was obtained from fermented tomatoes and identified on the basis of its cultural, morphological and biochemical characteristics20, followed by rDNA 16S gene analysis conducted at the Biomedical Research Center of La Rioja (CIBIR, Lab. of Genomics, Spain)21. Indicator microorganisms used in this study for the evaluation of the antimicrobial activity of the tested strain were isolated from various foods as well as outpatient pathological material.
Antibiogram of E. feacium: The bacterial culture (1.5×10 CFU mL–1) was inoculated on the Mueller-Hinton agar in the presence of 13 antibiotics and incubated at 30°C for 24 h. Resistance was defined as the lack of growth inhibit area around discs22.
Bacteriocin preparation: Enterococcus faecium was propagated at 30°C in MRS media23, collected after 12 h at the early stationary phase by centrifugation at 10.000 rpm for 20 min at 4°C. The cell free supernatant (CFS) was passed through membrane filters (Corning NY14831/Germany) with a pore diameter of 0.22 μm, neutralized to pH 6.5 (for bacteriocin like inhibitory substances analysis) and then stored at 4°C until use. Salmonella typhimurium (105 CFU mL–1) was used as an indicator strain in this study.
Antimicrobial spectrum: The agar well diffusion (AWD) assay and agar spot test were used to determine the antimicrobial spectrum of the test strain producing bacteriocin above mentioned and diameters of inhibition zones were scored22.
Effect of enzymes, heat, pH, UV light and storage time: The CFS was treated with enzymes (pepsin, trypsin, lipase and amylase, Oxford Laboratory Reagents), temperatures (120°C/15-20 min, 100°C/10 min, 75°C/10 min, 63-65°C for 30 min and 36-37°C/30-35 min), adjusted at pH (3, 3.5, 4, 4.5, 5, 5.5 and 6), exposed to UV irradiation (Herolab UV-4S/L, wave length 250 nm, Germany) for 5, 10, 15 and 20 min, stored at -20, 4 and 15°C for 15 days.
Effect of some food additives on bacteriocin activity: EDTA, citric acid, ascorbic acid, acetic acid, NaCl and sucrose were sterilized by filtration using bacterial filters porosity of 0.22 μm before use24.
Replacing MRS medium with agro-industrial wastes: Tomato and orange processing wastes were first boiled in water (100 g L–1) ), after decanting the solution obtained was sterilized by bacterial filters and used as growth media for E. faecium (2% v/v), the dairy whey was autoclaved for 10 min before use. The culture was tested for bacteriocin production using the optical density method ODM at 600 nm wavelength.
Statistical analysis: Data were expressed as mean±standard deviation. Statistical significance was determined using one-way analysis of variance on the replicates, where a p<0.05 was considered significant.
Fermented vegetables provide a good source for isolating LAB having antagonistic and probiotic properties. In this study, the search for bacteriocin production by some Enterococcus species using agro-based waste was undertaken. E. faecium produce an extraordinary array of microbial defense systems to maintain their existence or ecological niche against both bacteria and fungi. It is less common, which shows action against wide pathogens except Clostridium perfringens and Candida albicans (Table 1). The average diameter of inhibition zones as determined by the spot agar method ranged from 6-15 mm in size (Fig. 1 and 2).
| Table 1: | Antimicrobial spectrum of E. faecium |
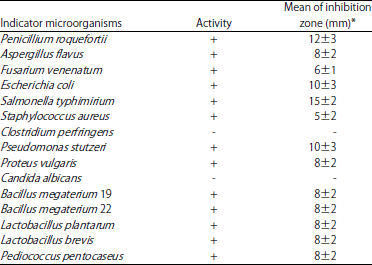 | |
+: Inhibition zone of size (mm), -: No inhibition zone, *Mean values (n = 3) of zone of inhibition include initial diameter of agar well. Values are Mean±SD | |
| Table 2: | Antibiotic susceptibility of E. faecium |
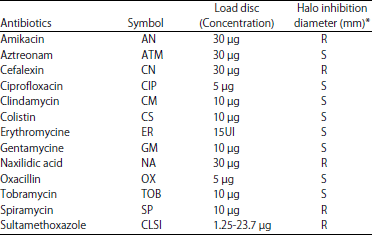 | |
*S: Sensible, R: Resistant | |
Antibiogram test showed that strain was highly resistant to amikacin, cefalexin, naxilidic acid, spiramycin and sultamethoxazole (Table 2) and sensitive to the rest antibiotics. These results reflected a complete inactivation of the antimicrobial activity after treatment of the supernatants with proteolytic enzymes, which reveals its proteinaceous nature and could be possibly added to feeds and foods.
No decrease in the activity was mentioned in the presence of NaCl and sucrose in compared with EDTA, citric acid, ascorbic acid and acetic acid. These results could be of industrial significance, as the bacteriocinogenic strain may be qualified for use in various food product applications based on its ability to stabilize the sensory quality and extend the shelf life of foods synergistically with additives included in the manufacture. Bacteriocin remained stable at pH values between 3.0 and 6.5, after heating at 120°C/15-20 min and exposing to UV light for 15 min (Table 3).
In another part of this research, different experiments were designed to attain the best available cultural conditions which could help in the production of bacteriocin, with the least economical costs possible and with the simplest methods available.
 | |
| Fig. 1(a-b): | (a) Morphological aspect and light micrographs of gram-stained cells of E. faecium. (b) Cells are gram-positive straight rods |
| Bar 12.5 = μm | |
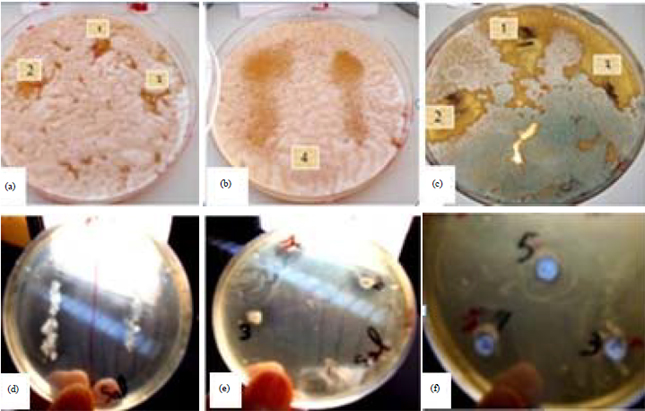 | |
| Fig. 2(a-f): | Antimicrobial activity of E. faecium against (a, b) Aspergillus flavus, (c) Penicillium roquefortii and (d-f) Salmonella typhimirium |
High bacteriocin production in this study was obtained with MRS broth within 6-12 h at 30°C, whey and tomato wastes 2 h at the beginning of the stationary growth phase, the low production levels recorded in oranges wastes (Fig. 3).
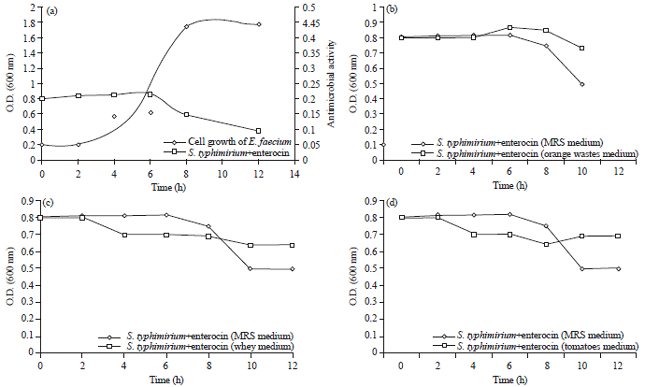 | |
| Fig. 3(a-d): | Bacterial growth and bacteriocin activity of E. faecium in (a) MRS, (b) Orange wastes, (c) Dairy whey and (d) Tomato wastes |
| Table 3: | Effect of pH, heat, UV light, storage temperature, enzymes and disinfectants agents on bacteriocin activity of E. faecium |
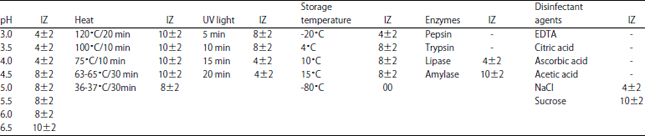 | |
IZ: Inhibition zone, +: Size of Inhibition zone (mm), –: No inhibition zone, *Mean values (n = 3) of zone of inhibition include initial diameter of agar well, Values are Mean±SD | |
Bacteriocins are highly specific antibacterial proteins produced by strains of bacteria active mainly against some other strains of the same or related species. The bacteriocins produced by LAB are potent biopreservative agents and the applications of these in food are currently the subject of extensive research5,11. In this study, the test strain E. feacium exhibited a broad antimicrobial spectrum, capable of being active against Gram positive and Gram negative representatives including food pathogens and fungi. These findings were already achieved by many researchers witch emphasized the effective role of enterococcal bacteriocins in foods and feeds6-19. E. faecium is naturally resistant to aminoglycosides, polymyxins and quinolones. It also has natural resistance to nitrofurans and sulfonamides. This resistance can be due to complex intrinsic features such as cell wall structure, metabolic properties or has been attributed as intrinsic and non-transmissible25-28. In an attempt to characterize the physical and biochemical properties of the bacteriocin produced by E. faecium strain, different factors such as heat, pH, storage, UV light, enzymes, additives were determined using S. typhimurium as indicator organisms29. The bacteriocin was stable in high temperature, acid and neutral pH, UV light exposition, refrigeration and freezing temperature. This is an interesting feature in view of its potential use as a food additive, in process like pasteurization, drying, tundalization, refrigeration and freezing, the resistance of bacteriocins in these range of temperatures make them effective against psychrophile, thermophile and mesophile food borne pathogens and also to different temperatures during production and storage. E. faecium bacteriocin characters make it effective in acidic and mesophilic environment, high and low temperature. Thermotolerance feature might be related to the molecular structure of the bacteriocin, usually composed by small peptides without tertiary structure. Treatment bacteriocin with disinfectant agents and non-ionizing artificial ultraviolet was to determine the synergistic or antagonistic interactions between this process for decontaminating surfaces of vegetables and other food products30-32. Good bacteriocin activity and production in commercial media such as MRS broth may suggest that specific nutrients are required for bacteriocin production. These results can be explained by the large amount of fermentable sugars in the tomato and lactose, proteins, vitamins and different mineral ions in dairy whey which make them good substrates for our bacteriocin production. Use of oranges wastes was not suitable due to pectin skins, limonene concentration and orange peel oil, which can inhibit bacteriocin secretion33-41.
Enterococcus faecium bacteriocin exhibited a wide antimicrobial spectrum and was capable of inhibiting the growth of many pathogens in foods products. The peculiar antimicrobial characteristics and technological properties and especially heat and storage stability and synergic effect with some foods additives, can positively have impact on their use as biopreservative, with a view to improve the hygiene and safety of the food products especially processed foods.
This study discovers the bacteriocins produced by lactic acid bacteria such as Enterococcus feacium that can be beneficial for foods and feeds biopreservation. This study will help the researcher to uncover the critical areas of the possibility of potential use of the bacteriocin as an antimicrobial agent to prevent the proliferation of some pathogenic and food spoilage microorganisms and to improve the microbial quality and safety of foods. Thus, new theories on foods biopreservation may be arrived at the replacement, the chemical preservatives by natural products.