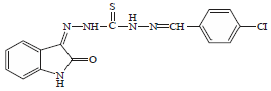Research Article
Antidiabetic Activity of 1-(4-Chlorobenzylidene)-5-(2-oxoindolin-3-ylidene) Thiocarbohydrazone in Chick Model
Jyothishmathi Institute of Pharmaceutical Sciences, Ramakrishna Colony, Thimmapur, 505481 Karimnagar, (Telangana State), India
Anreddy Rama Narsimha Reddy
Jyothishmathi Institute of Pharmaceutical Sciences, Ramakrishna Colony, Thimmapur, 505481 Karimnagar, (Telangana State), India
LiveDNA: 91.1516