ABSTRACT
The present study deals with the evaluation of phytoplankton community of Nigeen lake which is one of the rare aquatic ecosystem wherein the human population live inside the lake in house boats which serves also an attraction for high end tourism. Having this feature under consideration and also lack of proper sewage system connectivity of the house boats, it was thought worthwhile to have a pilot assessment of phytoplankton community in the lake. During the study period, a total of 65 phytoplankton taxa were identified representing four classes namely Chlorophyceae (28), Bacillariophyceae (24), Cyanophyceae (8) and Euglenophyceae (5). Chlorophyceae was found to be the dominant group in terms of diversity. However, the class wise representation depicted following order of dominance in terms of density of phytoplankton: Bacillariophyceae>Chlorophyceae>Cyanophyceae>Euglenophyceae. The percentage contribution of density also followed the same pattern with Bacillariophyceae contributing (50%), followed by Chlorophyceae (38%), Cyanophyceae (9%) and Euglenophyceae (3%) in a decreasing order. The most common genera encountered among all the sites included; Cymbella sp., Navicula sp., Tabellaria sp., Synedra sp., Amphora sp., Epithemia sp., Chlorella sp., Scenedesmus sp., Cosmarium sp., Tetraedron sp., Pediastrum sp., Oscillatoria sp., Anabaena sp., Phacus sp. and Trachelomonas sp. Among the sites studied, the highest value (3.45) of Shannon-Wiener diversity index was recorded at site II and lowest (3.24) at site V. The minimum value of Simpson diversity index (0.95) was recorded for site IV and a maximum of (0.96) was maintained at site II.
PDF Abstract XML References Citation
How to cite this article
URL: https://scialert.net/abstract/?doi=ajbs.2016.27.40
INTRODUCTION
Nutrient enrichment of lakes is one among the major environmental problems in many countries (Oczkowski and Nixon, 2008). Excess plant nutrients primarily nitrogen (N) and phosphorus (P) disposed into lakes mainly as untreated or partially treated domestic sewage, runoff from agricultural fields etc. stimulates the growth of algae and higher plants, which ultimately leads to the deterioration of water quality and degradation of entire ecosystem (Guyer and Ilhan, 2011; Luo et al., 2011). Since, a biotic community is the product of the interplay of myriad of factors and processes and interaction of different physical, chemical and geo-morphological characteristics of any water body, biological assessment in the form of phytoplankton is therefore a useful alternative in assessing these ecosystems (Stevenson and Pan, 1999).
Trophic status of inland water bodies is often evaluated on the information regarding the concentration of the limiting nutrient (phosphorus), chlorophyll (an indicator of phytoplankton biomass) and transparency (dependent on both algal biomass and sediment resuspension, expressed as Secchi depth). One of the most conspicuous and the best-known changes associated with eutrophication is the mass development of cyanobacteria, be it N2-fixing or nonfixing. Many species may develop toxic strains (e.g., M. aeruginosa, Cylindrospermopsis raciborskii ) and thus, large blooms may directly harm both other aquatic organisms and humans (Reynolds, 1990). Planktonic green algae include a large number of microscopic organisms, unicellular or colonial, adapted to spend part or all of their lives in apparent suspension in the open water of lakes, ponds and rivers. Plankton face a wide variation in availability of energy and materials among different lakes and within a given lake at different times and depths. This variation is matched by the different strategies employed in obtaining resources by different taxa, at a phylogenetic or functional group level and by the flexibility to exploit different types of resources depending on their relative availability (Elliott et al., 2000). The phytoplankton are important as ‘ecosystem engineers’ that alter the availability of the materials within an inland water body (Reynolds, 2006).
Phytoplankton have long been used as the indicators of water quality. Because of their short life span, they respond quickly to the environmental changes and hence species composition indicates the quality of the water mass in which they are found (Raven, 1982). The biological spectrum of the lentic fresh water bodies are multidimensional where phytoplankton is useful in bio-monitoring the ecological disturbance caused by a number of physico-chemical factors, sewage pollutants and other anthropogenic factors. The Nigeen lake from of Kashmir, which some of the researchers believe to be one of the basins of world famous Dal lake has become the worst victim of anthropogenic pressure in the last few decades. As a result of increasing population within the catchment area, direct discharge of untreated human wastes from houseboats and settlements adjoining the lake, human encroachments etc. that over the period of time has revealed signs of ecological deterioration including other aquatic systems (Bhat and Kamili, 2004; Zutshi and Ticku, 2006; Solim and Wanganeo, 2008; Ganie et al., 2010; Baba et al., 2011; Lone et al., 2011; Bhat, 2013; Parvez and Bhat, 2014). The rationale behind the present attempt was to actually study the dynamics of the phytoplankton community in relation to the increasing inputs of nutrients mainly in the form of autochthonous untreated sewage. It is with this background an attempt was made to study the species composition and population density of phytoplankton community of Nigeen lake.
MATERIAL AND METHODS
Study area: Nigeen lake, an offshoot leading from the Dal lake located at a distance of 9 km to the North-East of the Srinagar city India fall in the latitude of 34°7'13"N and longitude of 74°49'40"E with an altitude of 1584 m a.m.s.l., covering an area of 4.5 km2 (Rather et al., 2013). The lake is the main source of attraction for tourists and is exclusive in having number of houseboats, which provide excellent residing station for tourists and a source of income for lake dwellers. The houseboats are served by Shikaras (small boats) for transport and leisure. The lake is of the drainage type, being fed by a narrow water channel of Dal lake at Ashaibagh Bridge Sadiakadal in the North-East, while it drains into Gilsar and Khushalsar lake on its North-West side via Nallah Amir Khan. The water in Nigeen, in addition to being fed by Dal lake is also thought to be contributed by some springs within the lake bed and atmospheric precipitation. For the present investigation five sampling stations were selected to study the phytoplankton community of Nigeen lake (Fig. 1).
Description of study sites
Site I (Ashaibagh): This site is located at geographical coordinates of 34°06'53.0"N latitude and 74°50'09.5"E longitude near Ashaibagh Bridge in the North East side of Hazratbal basin receiving water from other basins of Dal lake and acts as an inlet source of Nigeen lake. This site serves as a parking station for Shikaras and is marked by the presence of floating gardens on one side. Submerged and free floating macrophytes are abundant.
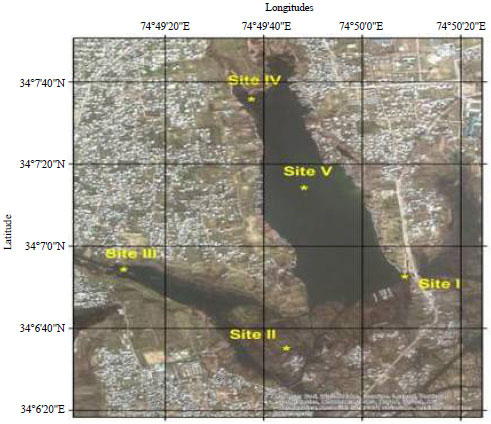 | |
| Fig. 1: | Satellite image of study area and study sites |
Site II (Khujyarbal): This site is located at geographical coordinates of 34°06'28.1"N latitude and 74°49'47.3"E longitude in the South West area of the lake. This site is marked by the presence of residential hamlets and is heavily impacted by the direct inflow of raw domestic sewage into the lake. It has proliferous growth of submerged and free floating macrophytes. Dense algal mats were also witnessed at this site.
Site III (Nallah Amir Khan): This site is located at geographical coordinates of 34°06'50.2"N latitude and 74°49'13.5"E longitude in the close proximity of the recently installed sewage treatment plant and acts as an outflow of Nigeen lake where from it drains into the Gilsar and Khushalsar lake. Submerged and free floating macrophytes are abundant.
Site IV (Bottabagh): This site is located at geographical coordinates of 34°07'37.33"N latitude and 74°49'38.2"E longitude towards the littoral zone of the lake. The site is marked by the presence of murky water harbouring rich algal growth.
Site V (Centre of Nigeen lake): This site is located at geographical coordinates of 34°07'14.2"N latitude and 74°49'49.3"E longitude near central deep zone of the lake with the maximum depth of about 6 m. This site is surrounded by a number of house boats and supports dense macrophytic vegetation.
The sampling was carried out during five months of the year, 2014. Two different approaches were opted for phytoplankton sampling for qualitative and quantitative estimations. Horizontal hauling was employed to a larger distance over the lake water at a particular site to get a statistically representative sample for qualitative purposes. Phytoplankton samples for quantitative analysis on the other hand were collected by sieving 100 L of water through a plankton net (No. 25, mesh size 64 μm) at different points at particular site in a composite manner. The plankton samples collected were then transferred to the properly labelled vials. Initially utmost care was given to identify the live phytoplankton. The samples were then preserved by fixing in 4% formalin (APHA., 1998). The aliquot in the tube was taken to be 50 mL. Identification of the phytoplankton was done with the help of standard works by Prescott (1939), Smith (1950), Edmondson (1992), Cox (1996), APHA (1998) and Biggs and Kilroy (2000). The quantitative estimation of phytoplankton was done under microscope by using Sedgwick Rafter cell of (1 mL capacity). The unicellular algae were counted as individuals where as 100 μm was taken as a unit in case of filamentous algae and in colonial forms like Volvox, Microcystis, Pandorina, Gomphosphaeria etc., the counting unit was a colony. Shannon and Wiener (1963), Simpson (1949) and Margalef (1951) indices were used to describe the numerical structure of the algal community. The species similarity between various sites was calculated by Sorenson similarity coefficient (Sorenson, 1948).
RESULTS AND DISCUSSION
In the present investigation, a total of 65 taxa of phytoplankton were identified. The study revealed that Chlorophyceae (28 taxa) and Bacillariophyceae (24 taxa) were the major contributors whereas, Cyanophyceae (8 taxa) and Euglenophyceae (5 taxa) were the least contributors (Table 1). However, the class wise representation depicted following order of dominance in term of density of phytoplankton: Bacillariophyceae>Chlorophyceae>Cyanophyceae>Euglenophyceae. The relative density of various groups at five different sites ranged between 48-55% (Baccillariophyceae), 31-43% (Chlorophyceae), 5-13% (Cyanophyceae) and 2-4% (Euglenophyceae) (Fig. 2). Based on the percentage contribution of different algal classes during whole study period, the algae belonging to Bacillariophyceae (50%) formed the bulk of phytoplankton followed by Chlorophyceae (38%), Cyanophyceae (9%) and Euglenophyceae ((3%) Fig. 3).
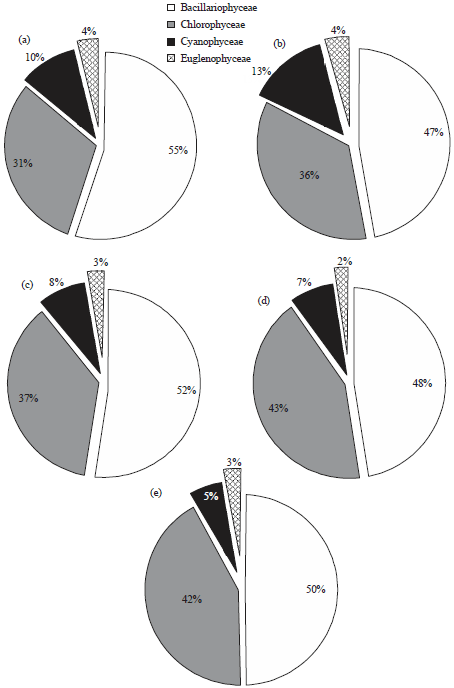 | |
| Fig. 2(a-e): | Relative density of different classes of phytoplankton at five different study sites |
The most abundant species among Bacillariophyceae in terms of mean population density were Cymbella sp., Navicula sp., Tabellaria sp., Fragilaria sp., Amphora sp. and Synedra sp., while Amphipleura sp., Surirella sp., Asterionella sp., Pinnularia sp. and Frustulia sp. were least abundant. In Chlorophyceae the most abundant species in terms of mean population density were Chlorella sp., Cosmarium sp., Scenedesmus sp., Pediastrum sp., Tetraedron sp. and Chlorococcum sp. and least abundant were Volvox sp., Closterium sp., Ankistrodesmus sp., Tribonema sp. and Rhizoclonium sp. Similarly, among Cyanophyceae, Anabaena sp. and Oscillatoria sp. were the most abundant in terms of mean population density, while Spirulina sp., Gomphosphaeria sp. and Calothrix sp. were the least abundant species. Euglenophyceae in the present study was the least represented group with five taxa only and among these Phacus sp. and Trachelomonas sp. were most abundant, while Lepocinclis sp., Euglena sp., Colacium sp. and were the least abundant species (Fig. 4).
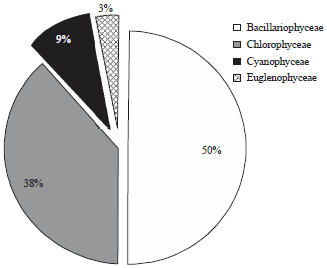 | |
| Fig. 3: | Percentage contribution of different classes of phytoplankton in Nigeen lake during entire study period |
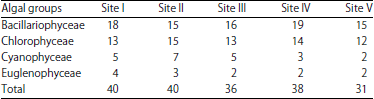
During the present study, the highest number of taxa were recorded each at site I and site II (40) followed by site IV (38), site III (36) and site V (31) (Table 1). The highest Shannon- Wiener diversity index was recorded for site II (3.45) followed by site I (3.37), site III (3.32), site IV (3.29) and the minimum was of site V (3.24). The minimum value of Simpson diversity index (0.95) was recorded for site IV and a maximum of (0.96) was maintained at site II. The maximum and minimum value of Margalef index was recorded for site II (4.34) and site V (3.81), respectively (Fig. 5). There was a lot of species similarity between different study sites as reflected by high Sorenson similarity coefficient (Fig. 6).
The phytoplankton community was not found to be so diverse. It may be due to samples collected from the lake were not of large enough volume to be representative of overall species distribution. Further, it is pertinent to mention here that there is thorough mixing of lake water in the Nigeen and as such plankton at the mercy of currents are distributed and scattered from one place to another. On the other hand, cell count of plankton, have been found to reach the mark of 30,000 individuals L–1, which is a clear sign of the impact of the human waste and untreated sewage entering into the lake.
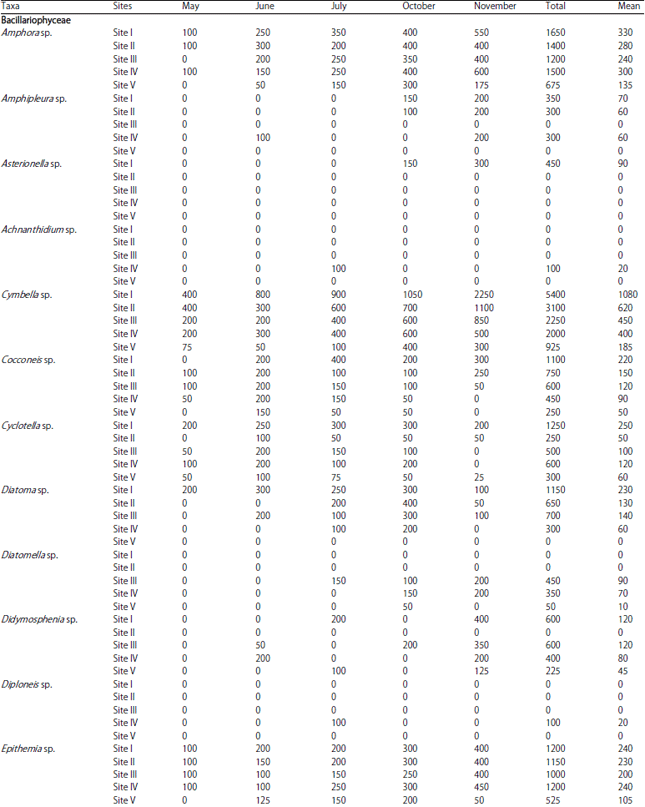
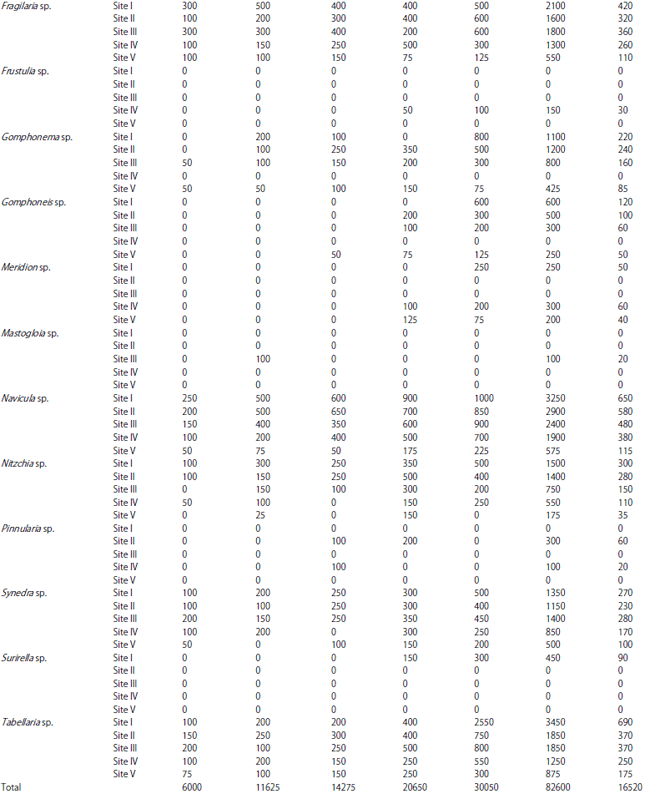
Phytoplankton communities do not respond only to natural changes into the lake but may also present variations as a consequence of human interventions affecting the water body, either directly or through activities carried on in the basin as a whole. These influences affecting the lake result in modifications to the structure and composition of the phytoplankton. In the present study, Bacillariophyceae formed the most dominant group of phytoplankton in terms of density. Bacillariophyceae constitutes the most important group of algae (Wetzel, 1983) and has been reported to be dominant among phytoplankton in lakes and wetlands by several authors (Mir and Kachroo, 1982; Zutshi and Wanganeo, 1984; Sarwar and Zutshi, 1987). The population density of Bacillariophyceae varied from a minimum of 450 individuals L–1 in May at site V to a maximum of 11700 individuals L‾1 in November at site I (Table 2).
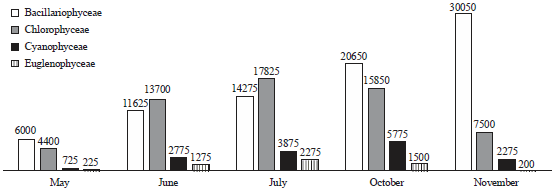 | |
| Fig. 4: | Monthly variation in total count of phytoplankton (individual L‾1) at five different sites during May to November, 2014 |
The highest population density of Bacillariophyceae or diatom group in the present investigation was recorded in November (Table 2) which could be attributed to the fact that they are able to grow under the condition of weak light and low temperature which are less suitable for other algae (Lund, 1965; Zafar, 1967; Goldman et al., 1968; Munawar, 1974; Ganai et al., 2010). Algal genera like Cymbella sp., Synedra sp., Fragilaria sp., Gomphonema sp., Cocconeis sp. and Navicula sp. which were recorded in the present lake are commonly found in organically polluted waters, thus indicating the organic load of the lake (Dickman and Kralina, 1975). The abundance of pollution tolerant taxa like Cymbella sp. and Navicula sp. at all the study sites indicates highest degree of organic pollution (Nandan and Aher, 2005).
Chlorophyceae formed the second most dominant group of phytoplankton. Some genera of green algae like Scenedesmus sp., Pediastrum sp., Coelastrum sp., Tetraedron sp. etc. have been reported to be abundant in eutrophic waters (Hutchinson, 1967). Most of these genera were found in the present lake and as such formed greater proportion of the green algae plankton. Taxa like; Coelastrum, Oocystis, Scenedesmus, Pandorina, Pediastrum, Zygnema, Chlorella, Tetraedron, Spirogyra and Tribonema have been reported to thrive in nutrient rich environs and majority of these green algae were recorded in the lake under study. The population density of Chlorophyceae reached its highest peak (4650 individuals L–1) at site I in July, while as the lowest population density (400 individuals L–1) was obtained at site V in May. During the study period, Chlorophyceae showed its peak value in July (Table 2). This could be attributed to the increase in temperature in addition to the increased P and N concentration (Kant and Kachroo, 1977; Ganai et al., 2010).
Cyanophyceae formed the third most dominant group of phytoplankton. In current study, occurrence of Microcystis at site I, site II, site III and site IV, which is considered as an indicator of eutrophic conditions (Rawson, 1956) may be attributed to the highest degree of civic pollution at these study sites (Nandan and Aher, 2005).
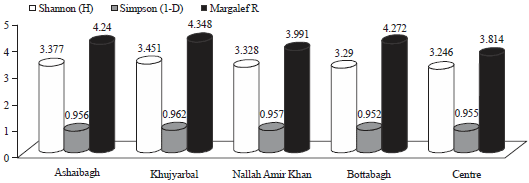 | |
| Fig. 5: | Diversity indices of phytoplankton at five different study sites |
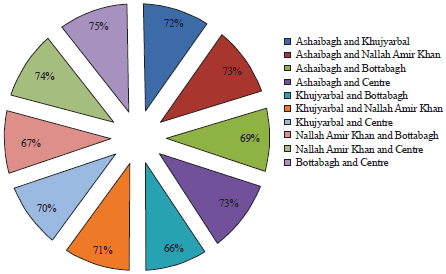 | |
| Fig. 6: | Serenson’s similarity coefficient of phytoplankton at different five study sites |
| Table 1: | Monthly variations in population density (ind./l) of phytoplankton at different sites |
  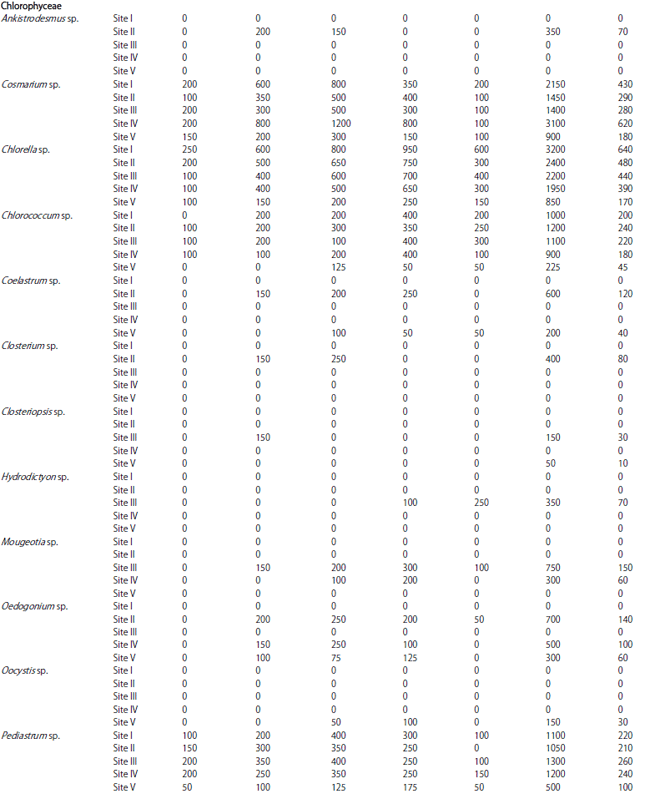 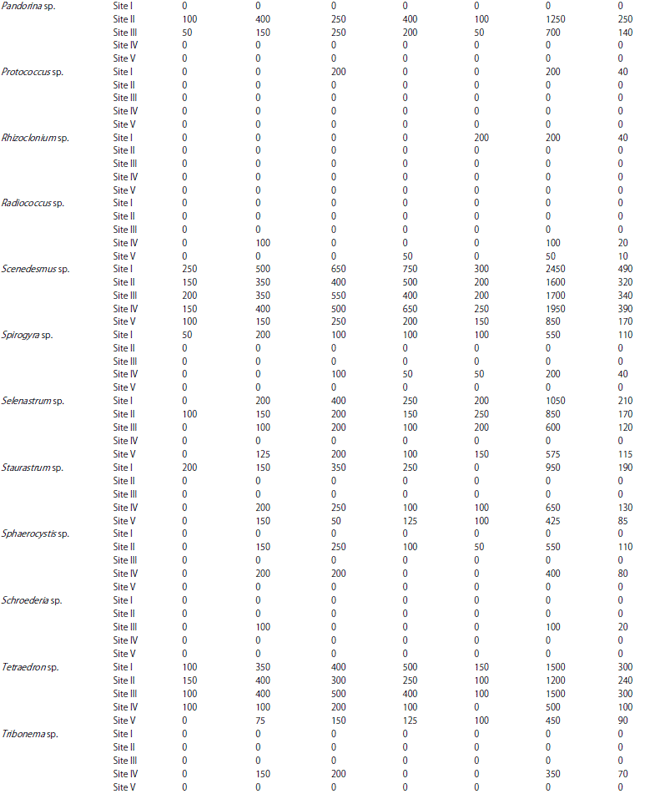 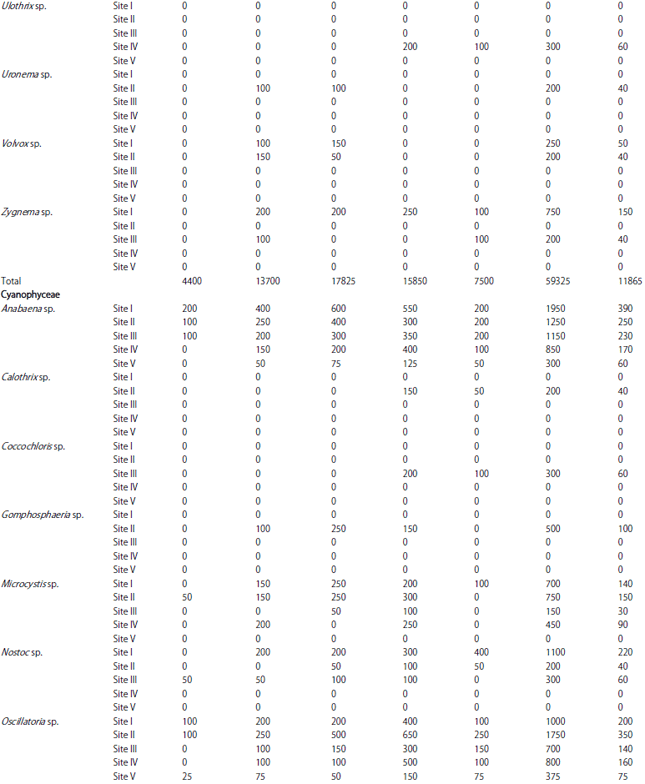 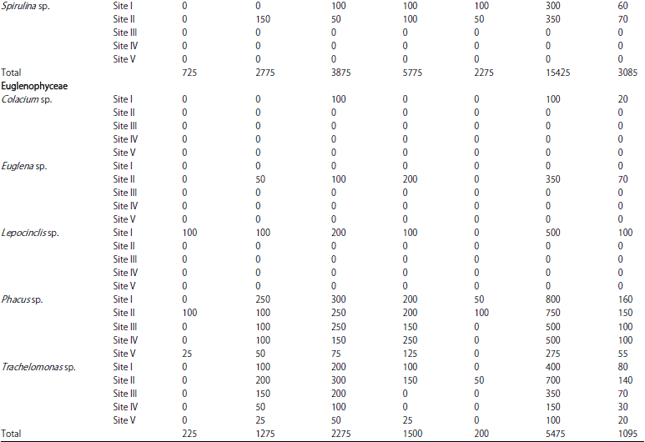 | |
| Table 2: | Number of phytoplankton taxa at five different study sites |
 | |
Further, the presence of Oscillatoria at all the study sites indicates pollutants of biological origin, which agrees with the observations of Gadag et al. (2005). The above study gains further support from the studies of Kumar (1990) and Zargar and Ghosh (2006) that excessive growth of certain algal genera viz., Anabaena and Oscillatoria is an indicative of nutrient enrichment of aquatic bodies. Among the sites studied the percentage composition of Cyanophyceae (13%) has been found to be higher at site II (Khujyarbal), which is marked by the presence of human settlements, where water is contaminated with sewage and faecal matter. Pearsall (1932) and Sedamkar and Angadi (2003) had correlated abundance of Cyanophyceae with high concentration of organic matter and nutrients.
Euglenophyceae in the present study formed the least represented group of phytoplankton and showed its peak development in July (Table 2), which can be attributed to the increasing temperature in addition to the level of organic matter (Hutchinson, 1967). Among the sites studied, Euglena was recorded only at site II indicating significant quantities of organic matter at this site, which may be attributed to the direct discharge of sewage from nearby human settlements. According to Hutchinson (1967), Euglena and Phacus are characteristics of waters having significant quantities of organic matter. The presence of both the taxa in the lake under study indicates the water body to be rich in organic matter.
Nigeen lake is subjected to pollution due to addition of fertilizers from floating gardens and domestic sewage from human habitation. Progressive enrichment of water with nutrients leads to mass production of algae, which in turn leads to the increased productivity and other undesirable biotic changes (Ahmad, 1996). A number of researcher have reported many algal species as indicators of water quality (Naik et al., 2005; Nandan and Aher, 2005; Zargar and Ghosh, 2006). Nandan and Aher (2005) has shown the algal genera, like Euglena, Oscillatoria, Scenedesmus, Navicula, Nitzschia and Microcystis to flourish in organically polluted waters. This almost comborates with the earlier study by Palmer (1969) who maintained that genera like Scenedesmus, Oscillatoria, Microcystis, Navicula, Nitzschia and Euglena mostly belonging to organically polluted waters. The very much presence of above taxa in the present water body suggests the lake to be organically polluted.
CONCLUSION
The Phytoplankton contribution towards the productivity in terms of cell number was mostly from Chlorophyceae and Bacillariophyceae with very minor contribution of less than 15% from Cyanophyceae and Euglenophyceae, which has been a usual trend in case of majority of Valley lakes. Chlorophyceae and Bacillariophyceae were more or less found equally in terms of species composition, while as the Cyanophyceae and Euglenophyceae contributed less number of phytoplankton in the Nigeen lake during the study period. The cell counts as found in the lake were enormously high thereby indicating the visible impact of sewage on the growth and multiplication of phytoplankton in the lake. The thick mats of algae dominated by Oscillatoria were found near site II and V possibly triggered by excessive enrichment from sewage of surrounding residential hamlets and houseboats. The signs of the floating of human waste, foul smell possibly due to hydrogen sulphide, decomposition of organic matter in the form of macrophytes but dominated by algal mats, anoxic conditions of the water were very visible during the sample visits at these sites. Since, there are various variables affecting the growth and multiplication of phytoplankton and therefore the species dynamics must be carefully evaluated and monitored further to reach on some concrete conclusions.
ACKNOWLEDGMENTS
The authors are highly thankful to H.O.D Environmental Science, University of Kashmir for providing the necessary laboratory facilities to carry out this work. Further, the help rendered by research scholars of Aquatic Ecology Lab namely Showkat Ahmad, Adil Hamid and Inam Sabha is greatly acknowledged.
REFERENCES
- Baba, A.I., A.H. Sofi, S.U. Bhat and A.K. Pandit, 2011. Periphytic algae of River Sindh in the Sonamarg area of Kashmir valley. J. Phytol., 3: 1-12.
Direct Link - Bhat, T.H., 2013. Physicochemical characteristics of Dal lake under temperate conditions of Kashmir (Jammu and Kashmir), India. Int. J. Curr. Sci., 9: 133-136.
Direct Link - Elliott, J.A., A.E. Irish, C.S. Reynolds and P. Tett, 2000. Modelling freshwater phytoplankton communities: An exercise in validation. Ecol. Modell., 128: 9-16.
CrossRefDirect Link - Luo, G., F. Bu, X. Xu, J. Cao and W. Shu, 2011. Seasonal variations of dissolved inorganic nutrients transported to the Linjiang Bay of the three Gorges reservoir, China. Environ. Monit. Assess., 173: 55-64.
CrossRefDirect Link - Guyer, G.T. and E.G. Ilhan, 2011. Assessment of pollution profile in Buyukcekmece Watershed, Turkey. Environ. Monitor. Assess., 173: 211-220.
CrossRefDirect Link - Ganai, A.H., S. Parveen, A.A. Khan and H. Maryam, 2010. Phytoplankton diversity at Watlab Ghat in Wular lake, Kashmir. J. Ecol. Nat. Environ., 2: 140-146.
Direct Link - Jalal, K.C.A., B.M.A. Azfar, B.A. John and Y.B. Kamaruzzaman, 2011. Spatial variation and community composition of phytoplankton along the pahang estuary, Malaysia. Asian J. Biol. Sci., 4: 468-476.
CrossRef - Lone, S.A., A.K. Pandit and S.U. Bhat, 2011. Dynamics of periphytic algae in some crenic habitats of district Anantnag, Kashmir. J. Himalayan Ecol. Sustian. Dev., 7: 28-34.
Direct Link - Lund, J.W.G., 1965. The ecology of the freshwater phytoplankton. Biol. Rev., 40: 231-290.
CrossRefDirect Link - Mir, A.M. and P. Kachroo, 1982. Limnology of Kashmir lakes. VII: The ecology of Bacillariophyceae in two lakes of Srinagar. Proc. Indian Natl. Acad., 48: 378-390.
Direct Link - Munawar, M., 1974. Limnological studies on freshwater ponds of Hyderabad, India IV. The biocenose. Periodicity and species composition of unicellular and colonial phytoplankton in polluted and unpolluted environments. Hydrobiologia, 45: 1-32.
CrossRefDirect Link - Nandan, S.N. and N.H. Aher, 2005. Algal community used for assessment of water quality of Haranbaree dam and Mosam river of Maharashtra. J. Environ. Biol., 26: 223-227.
PubMed - Oczkowski, A. and S. Nixon, 2008. Increasing nutrient concentrations and the rise and fall of a coastal fishery; a review of data from the Nile Delta, Egypt. Estuarine Coastal Shelf Sci., 77: 309-319.
CrossRefDirect Link - Palmer, M.C., 1969. A composite rating of algae tolerating organic pollution. J. Phycol., 5: 78-82.
CrossRefDirect Link - Parvez, S. and S.U. Bhat, 2014. Searching for water quality improvement of Dal Lake, Srinagar, Kashmir. J. Himalayan Ecol. Sustain. Dev., 9: 51-64.
Direct Link - Pearsall, W.H., 1932. Phytoplankton in the English lakes: II. The composition of the phytoplankton in relation to dissolved substances. J. Ecol., 20: 241-262.
CrossRefDirect Link - Raven, J.A., 1982. The energetics of freshwater algae; Energy requirements for biosynthesis and volume regulation. New Phytol., 92: 1-20.
CrossRefDirect Link - Reynolds, C.S., 1990. Temporal scales of variability in pelagic environments and the response of phytoplankton. Freshwater Biol., 23: 25-53.
CrossRefDirect Link - Solim, S.U. and A. Wanganeo, 2008. Excessive phosphorus loading to Dal Lake, India: Implications for managing shallow Eutrophic Lakes in urbanized watersheds. Int. Rev. Hydrobiol., 93: 148-166.
CrossRefDirect Link - Sorenson, T., 1948. A method of establishing groups of equal amplitude in plant sociology based on similarity of species and its application to analyses of the vegetation on Danish commons. Biol. Skrifter, 5: 1-34.
Direct Link - Zutshi, D.P. and A. Ticku, 2006. Impact of mechanical deweeding on Dal Lake ecosystem. Dev. Hydrobiol., 61: 419-426.
CrossRefDirect Link - Zafar, A.R., 1967. On the ecology of algae in certain fish ponds of Hyderabad, India. III. The periodicity. Hydrobiologia, 30: 96-112.
CrossRefDirect Link - Zargar, S. and T.K. Ghosh, 2006. Influence of cooling water discharges from Kaiga nuclear power plant on selected indices applied to plankton population of Kadra reservoir. J Environ. Biol., 27: 191-198.
Direct Link








