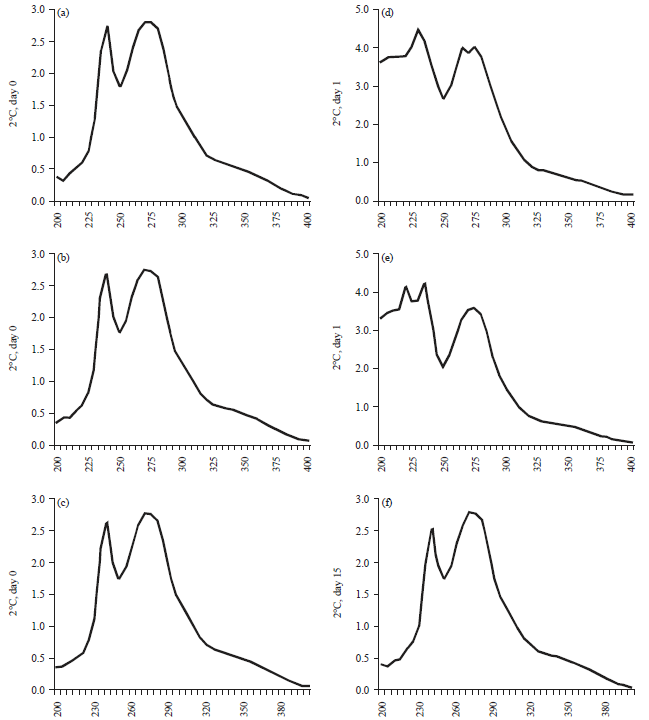
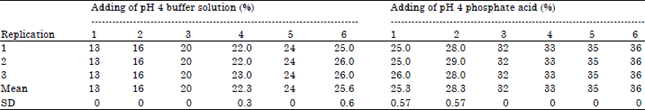
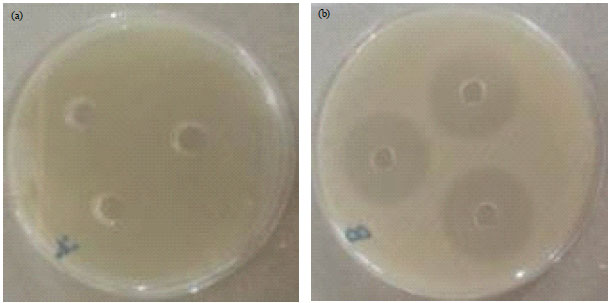
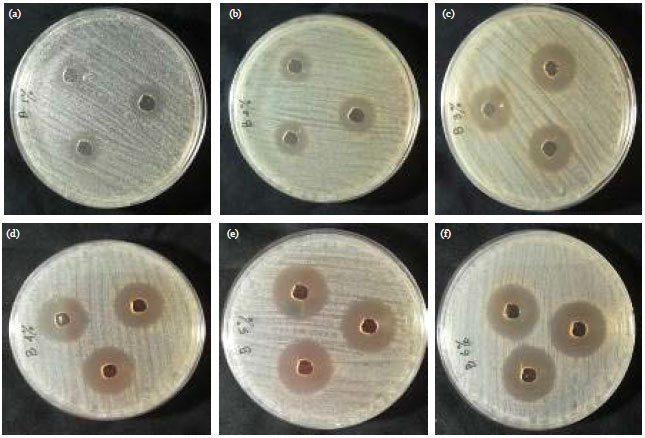
Research Article
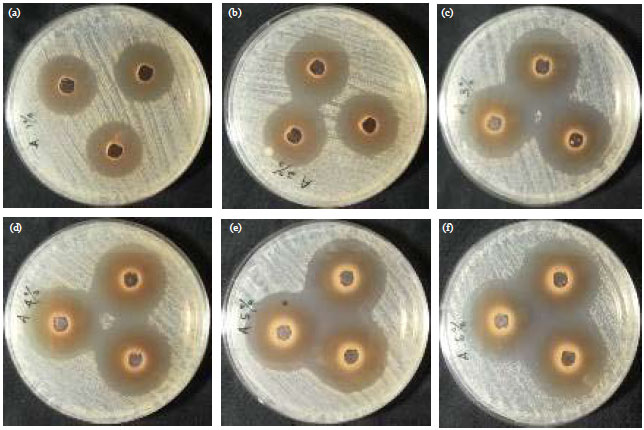
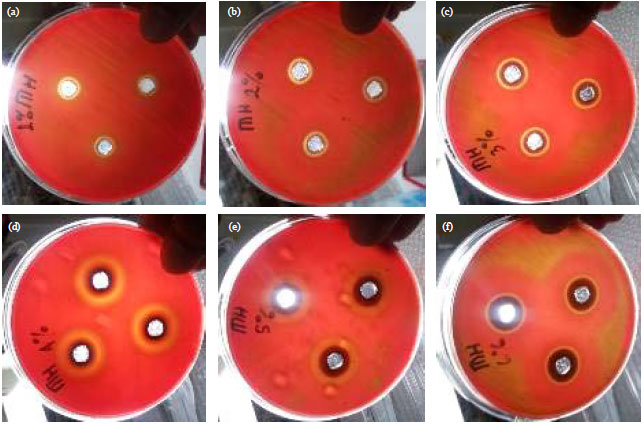
Stability of Epigallocatechin Gallate (EGCG) from Green Tea (Camellia sinensis) and its Antibacterial Activity against Staphylococcus epidermidis ATCC 35984 and Propionibacterium acnes ATCC 6919
Faculty of Pharmacy, Gadjah Mada University, Indonesia
Achmad Fudholi
Faculty of Pharmacy, Gadjah Mada University, Indonesia
Sudarsono
Faculty of Pharmacy, Gadjah Mada University, Indonesia
Erna P. Setyowati
Faculty of Pharmacy, Gadjah Mada University, Indonesia