Research Article
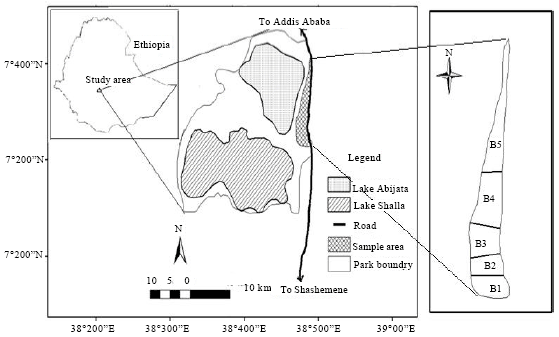
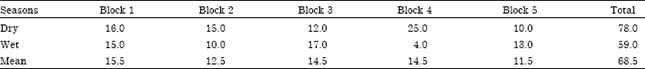
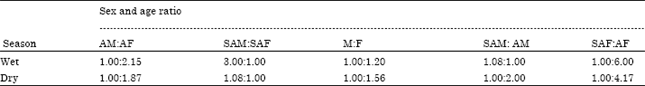
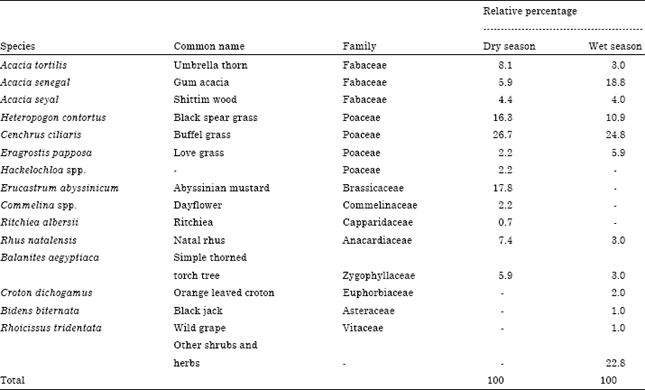
Population Status, Feeding Ecology and Activity Patterns of Grant’s Gazelle (Gazella granti) in Abijata-Shalla Lakes National Park, Ethiopia
Department of Biology, Debre Markos University, P.O. Box 269, Debre Markos, Ethiopia
Afework Bekele
Fucalty of Life Sciences, Addis Ababa University, P.O. Box 1176, Addis Ababa, Ethiopia