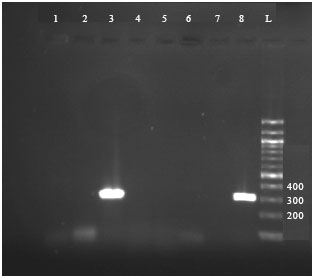
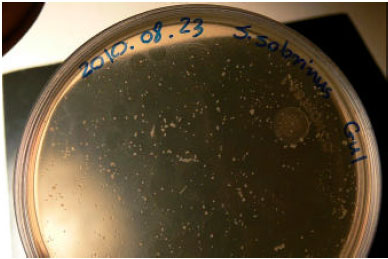
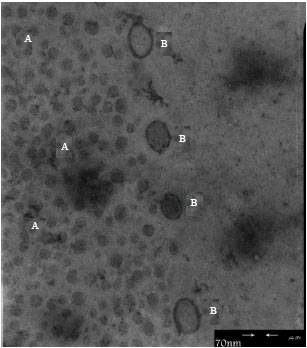
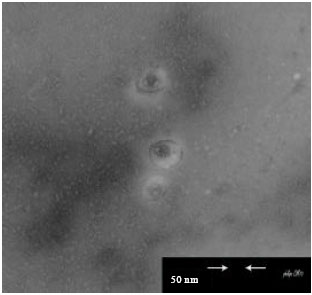
Research Article
Characterization of Two Lytic Bacteriophages of Streptococcus sobrinus Isolated from Caspian Sea
Department of Biology, Faculty of Science, University of Isfahan, Isfahan 81746-73441, Iran
Majid Bouzari
Department of Biology, Faculty of Science, University of Isfahan, Isfahan 81746-73441, Iran
Farahnaz Arbabzadeh Zavareh
Department of Operative Dentistry, School of Dentistry, Isfahan University of Medical Sciences, Isfahan 81746-73461, Iran