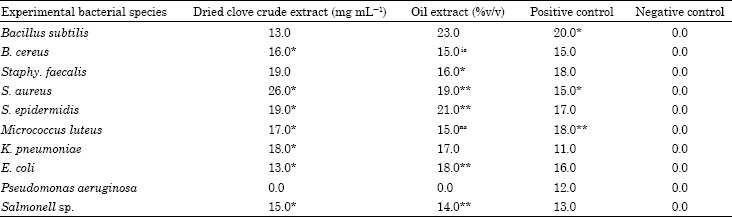
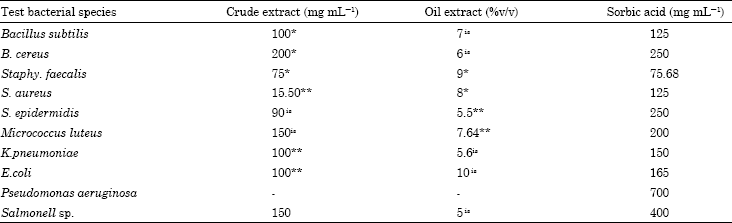
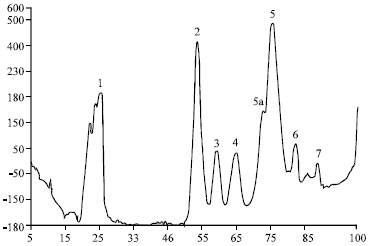
Research Article
Bioactive Compounds and its Autochthonous Microbial Activities of Extract and Clove Oil (Syzygium aromaticum L.) on Some Food Borne Pathogens
International Centre for Bioresources Management, Malankara Catholic College, Mariagiri, Kaliakkavilai, 629153, Kanyakumari District, Tamil Nadu, India
S. Sujatha
International Centre for Bioresources Management, Malankara Catholic College, Mariagiri, Kaliakkavilai, 629153, Kanyakumari District, Tamil Nadu, India