Research Article
A Histological and Ultrastructural Study of Gland Cells in the Ovary of the Sexually Immature Ostrich (Struthio camelus)
Department of Veterinary Anatomy, Sokoine University of Agriculture, P.O. Box 3016, Morogoro, Tanzania
The presence of gland cells in the ovary of birds has been extensively reported (Marshall and Coombs, 1957; Dahl, 1970; Erpino, 1973; Guraya and Chalana, 1976; Chalana and Guraya, 1979; Parizzi et al., 2008; Rueda-Cediel et al., 2008). According to Gilbert (1979), the origin and importance of gland cells in the avian ovary is still controversial. Marshall and Coombs (1957) reported the occurrence of gland cells in the atretic ovary of the rook (Corvus frugilegus), which were thought to have originated from fibroblasts in the thecal layer. In contrast, Erpino (1973) reported the presence of gland cells in the ovarian stroma which were thought to have originated from thecal cells of atretic follicles, as well as from stromal fibroblasts.
Knowledge of the biology of gland cells in the ovary of birds is important, not only due to the functional significance of the gland cells, but also due to the fact that the structural features of these gland cells appears to vary depending on the location within the ovary. A report by Guraya (1976) shows that gland cells in the ovaries of the pigeon (Columba livia) and ring dove (Streptopelia decaocto) plays an important role on steroidogenesis. To date much of the information regarding the structure and function of gland cells in the ovary has been restricted to the domestic fowl (Dahl, 1970, 1971; Guraya, 1989; Velazquez et al., 1991; Forgo et al., 1988). There is a paucity of information on the presence and distribution of gland cells in the ovary of the sexually immature ostrich. This study was therefore undertaken to describe the morphological features of gland cells in the ovary of the sexually immature ostriches.
A total of 26 sexually immature female ostriches aged between 12-14 months old were used in the present study. The birds were sacrificed by stunning followed by decapitation at a commercial ostrich abattoir in the Republic of South Africa in 2009. Ovarian tissue samples were collected as soon as possible following the death of the bird. The tissue samples for light microscopic study were fixed in 10% buffered formalin and processed routinely using an automated tissue processor (Shandon Excelsior, Thermo electron Corporation, Germany) and stained with Haematoxylin and Eosin (Drury and Wallington, 1976). Tissue samples for transmission electron microscopy were immersion-fixed in 2.5% glutaraldehyde in 0.1 M cacodylate buffer. After post-fixation with osmium tetroxide (OsO4) for 1-2 h, the tissue blocks were rinsed with cacodylate buffer for 10 min and distilled water for a further 10 min. Thereafter, the tissue sections were dehydrated using a graded series of alcohol (50-100% ethanol). Tissue sections were infiltrated with propylene: epoxy resin mixture at a ratio of 2:1, followed by a 1:1 for 1 h and then embedded in a 100% epoxy resin overnight. The tissue sections stained with lead citrate and uranyl acetate and viewed with a transmission electron microscope (Philips CMIO).
Histological observation: Thecal gland cells were observed in the thecal layer of both pre-vitellogenic and vitellogenic follicles. Two types of thecal gland cells were identified, refereed here as type I and II thecal gland cells. Type I are undifferentiated whilst the type II are differentiated thecal gland cells. Type I thecal gland cells contained oval nuclei surrounded by scant cytoplasm. Type II thecal gland cells contained centrally-located round nuclei enclosed with lightly stained cytoplasm (Fig. 1). Although, the gland cells were evenly distributed in the thecal layers, theca externa appeared to contain more type I thecal gland cells than type II.
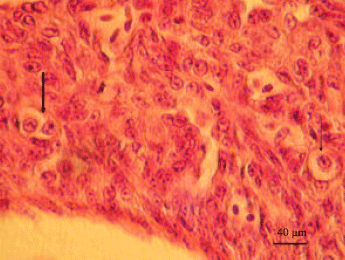 | |
| Fig. 1: | Photomicrograph of thecal layer in the late pre-vitellogenic follicle of the sexually immature ostrich. Individual (thin arrow) and group (thick arrow) of thecal gland cells are observed. The gland cells display abundant cytoplasm which stained lightly with H and E |
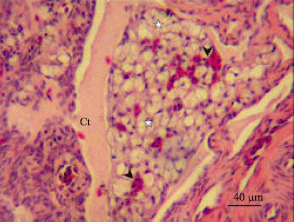 | |
| Fig. 2: | Photomicrograph of ovarian stroma in the sexually immature ostrich. A group of interstitial gland cells (asterisks) are observed. Blood vessels (arrowheads) are seen interspaced between the glands. A connective tissue band (Ct) is seen enclosing the glands |
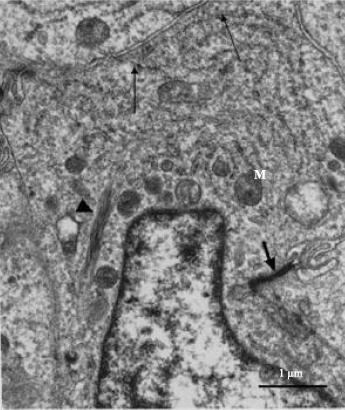 | |
| Fig. 3: | Electron photomicrograph of type I thecal gland cell in the thecal layer of pre-vitellogenic follicle in the ovary of the sexually immature ostrich. Cisternae of RER (arrows), mitochondria (M) and filamentous bundle (arrowhead) are observed in the cytoplasm. The cytoplasm contains materials of an intermediate electron density. Cellular junction (short arrow) is observed along the lateral plasma membrane. N: Nucleus |
Intertitial glands, which originated from theca interna cell, were observed in the theca interna and externa of atretic vetillogenic follicles. The interstitial gland cells were observed in groups, occupying major portion of the thecal layer (Fig. 2). The gland cell contained round nucleus surrounded by scant cytoplasm.
Ultrastructural observation: Type I thecal gland cells contained large oval-shaped or elongated nuclei, which exhibited clumps of heterochromatin. The cytoplasm of these cells contained particles of an intermediate electron density. Relatively few organelles such as: mitochondria and rough endoplasmic reticulum were observed throughout the cytoplasm (Fig. 3). Filamentous bundles were identified perinuclearly (Fig. 3). Type II thecal gland cells were observed in the thecal layer of pre-vitellogenic and vitellogenic follicles. The type II gland cells occurred either singly or in groups of 3 to 6 cells (Fig. 4a). A relatively thin fibrous tissue surrounded the group of thecal gland cells (Fig. 4a). Within the group, tight junctions linked the adjacent gland cell (Fig. 4b). The type II thecal gland cells contained centrally-located round to oblong nuclei with prominent nucleoli (Fig. 5). The cytoplasm of these cells was electron dense with numerous lipid droplets (Fig. 4a, 5).
Interstitial gland cells were distributed in groups in both theca interna and theca externa of atretic vitellogenic follicles. In early stages of atresia, interstitial cells contained eccentrically placed heterochromatic nuclei (Fig. 6). The cytoplasm of these cells was filled with lipid droplets which obscured the visibility of cytoplasmic organelles. Scant cytoplasm was observed between the lipid droplets. In advanced stages of atresia, several of the interstitial gland cells appeared to undergo degeneration. At this stage, the cells contained pyknotic nuclei and a few lipid droplets. In addition, cellular debris was present in the areas between the gland cells with few macrophages infiltration occasionally identified (Fig. 6).
The result of the current investigation has highlighted the morphology of the gland cells in the ovary of the sexually immature ostrich. This appears to be the first account on the presence of gland cells in the sexually immature ostrich. Earlier reports on the morphology of the gland cells have mainly been confined to the adult bird (Dahl, 1970, 1971; Guraya, 1989; Velazquez et al., 1991).
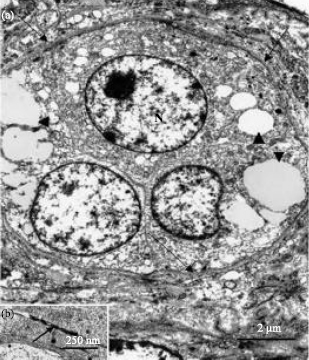 | |
| Fig. 4: | (a) Electron photomicrograph of a group of type II thecal gland cells in the thecal layer of vitellogenic follicle in the ovary of the sexually immature ostrich. The gland cells contain round nuclei (N). Lipid droplets (arrowheads) of variable sized are observed in the cytoplasm. Note the presence of a fibrous connective tissue (arrows) enclosing the thecal glands and (b) A higher magnification electron photomicrograph of a tight junction (arrow) joining adjacent gland cell |
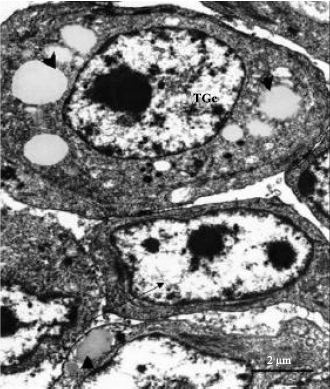 | |
| Fig. 5: | Electron photomicrograph of type I (arrow) and type II (TGc) thecal gland cells in the thecal layer of pre-vitellogenic follicle in the ovary of the sexually immature ostrich. Note the presence of numerous lipid droplets (arrowheads) and centrally-located round nucleus in the type II gland cell. The nucleus in the type I is oval-shaped surrounded by scant cytoplasm |
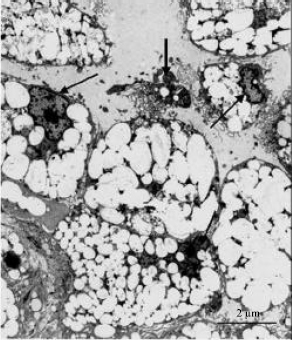 | |
| Fig. 6: | Electron photomicrograph of interstitial glands in the thecal layer of atretic vitellogenic follicle in the ovary of sexually immature ostrich. The gland cells contain numerous lipid droplets (asterisks) and eccentrically-located nuclei (arrows). Note the presence of cellular debris (thick arrow) adjacent degenerating gland cell |
This could be due to the fact that reproductive tract in birds become fully functional after reaching puberty.
Different names have been used to describe gland cells in the avian ovary. The used names include: interstitial glands (Chalana and Guraya, 1979), stromal glands (Erpino, 1973), as well as, steroid producing and enclosing cells (Dahl, 1970). In the present study, the term thecal and interstitial gland cells were used based on their location in the thecal layer, as well as, in the ovarian stroma.
As mentioned earlier, gland cells have been shown to originate from various ovarian cells (Marshall and Coombs, 1957; Erpino, 1973; Dahl, 1970). In the present investigation, thecal gland cells appeared to be formed from undifferentiated thecal cells. They started to appear in the thecal layer during late pre-vitellogenic phase. Some thecal gland cells were seen either singly or in groups. Similar observation was also made in the ovary of the house sparrow (Guraya and Chalana, 1976), crow and myna (Chalana and Guraya, 1979). In these species thecal gland cells were distributed either in groups or singly in the ovarian stroma.
At light microscopic level, gland cells displayed abundant cytoplasm which stained lightly with H&E. Similar finding was observed in the ovaries of the rat (Dahl, 1971). In the rat, gland cells contained round nucleus surrounded by clear cytoplasm. Transmission electron microscopic observations of both type I and type II thecal gland cells revealed abundant cytoplasm, which contained numerous lipid droplets, mitochondria and filamentous bundles. This observation suggests that thecal gland cells in the sexually immature ostrich could be involved in the synthesis of steroid hormones. In support of this idea, Velazquez et al. (1991) and Hernandez-Vertiz et al. (1993) described the structure of steroidogenic cells in the thecal layer of the ovary of the domestic fowl (Gallus domesticus). The reports showed that a typical steroidogenic cell is characterized by abundant lipid droplets in the cytoplasm.
In addition to thecal gland cells, interstitial gland cells were observed in the thecal layer and stroma of atretic ovarian follicles. Similar findings were also observed in the ovaries of house sparrow (Guraya and Chalana, 1976), pigeon and dove (Guraya, 1976), as well as, scrab jay (Erpino, 1973) during atresia. According to these reports, interstitial gland cells were prominent during breeding season. It was proposed that interstitial gland cells serve as lipid reservoir for the synthesis of steroid hormone during breeding season. This can also be the case in the current study. In the sexually immature ostrich the ovary undergoes cycles of activity and in activity (Madekurozwa and Kimaro, 2006). During the active phase, the ovarian follicles developed to the vitellogenic stage and then regressed.
In conclusion, the present description of the morphology of the thecal and interstitial gland cells warrants further investigations on the functional analysis of these cells in the ovary of sexually immature ostrich. Although, it is undoubted that thecal gland cells in the avian species participate in steroid hormone secretion, no investigation appears to have been carried out to ascertain this and the nature of their steroid hormone.
The Deutscher Akademischer Austauschdienst (DAAD-ANSTI program) and University of Pretoria funded this study. The author thank staff of the electron microscope unit and histopathology laboratory of the University of Pretoria for their technical assistance.