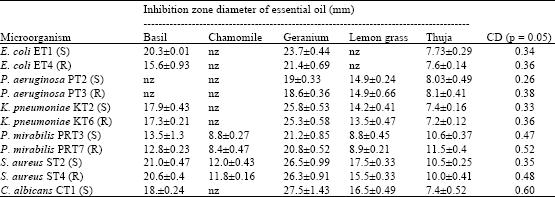
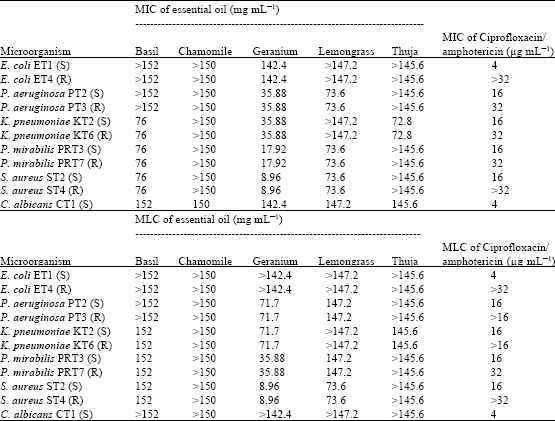
Research Article
Antimicrobial Effects of Essential Oils against Uropathogens with Varying Sensitivity to Antibiotics
Department of Microbiology, Kanya Gurukul Mahavidyalaya, Gurukul Kangri University, Jwalapur, Hardwar-249407, India
Padma Singh
Department of Microbiology, Kanya Gurukul Mahavidyalaya, Gurukul Kangri University, Jwalapur, Hardwar-249407, India