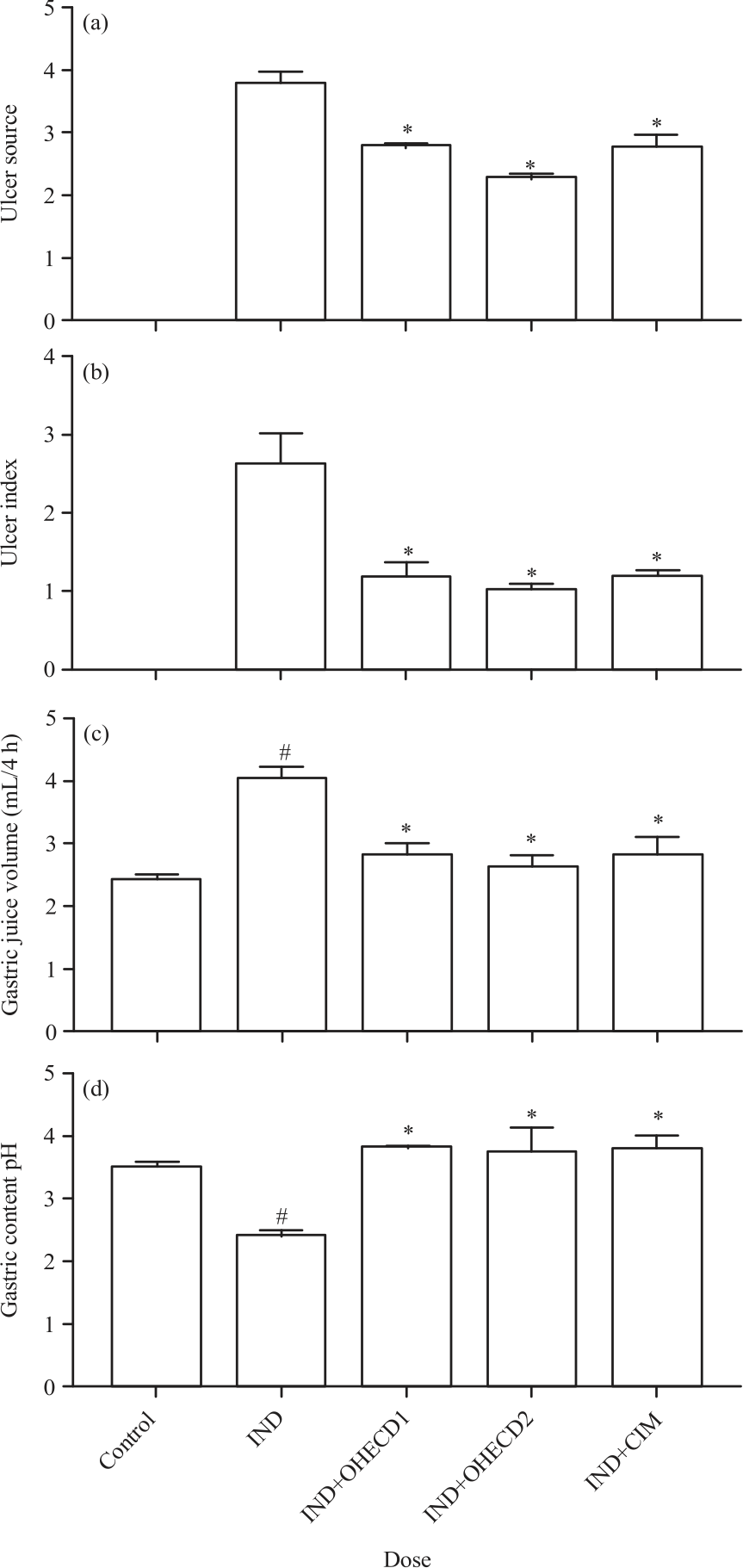
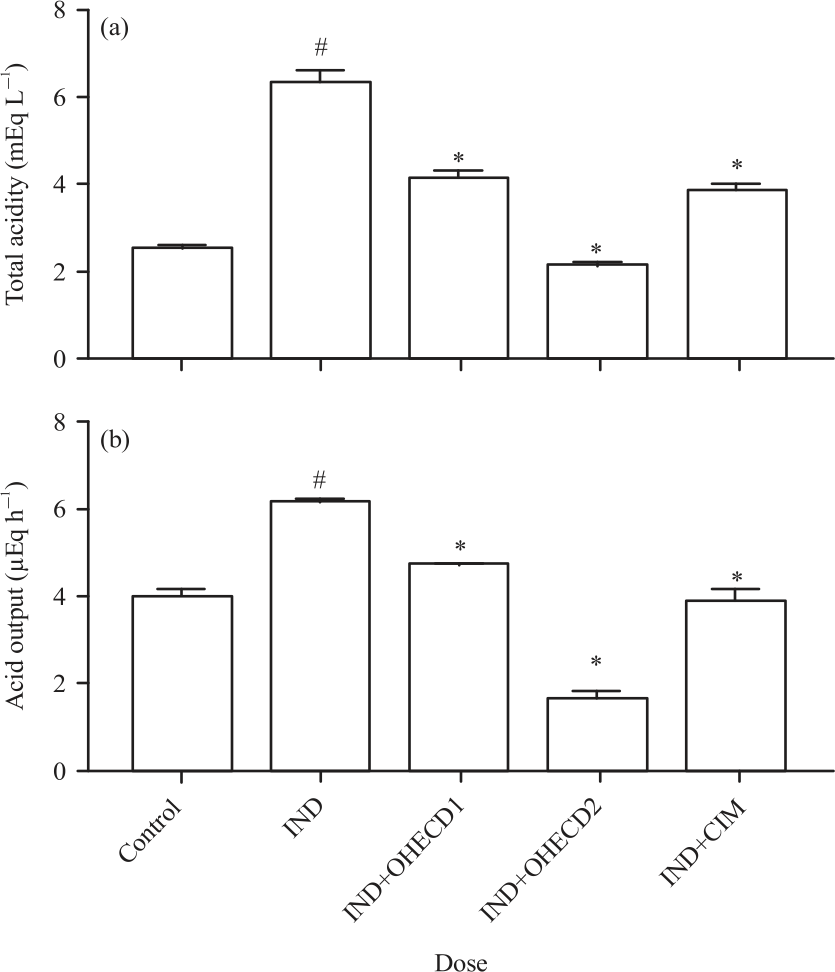
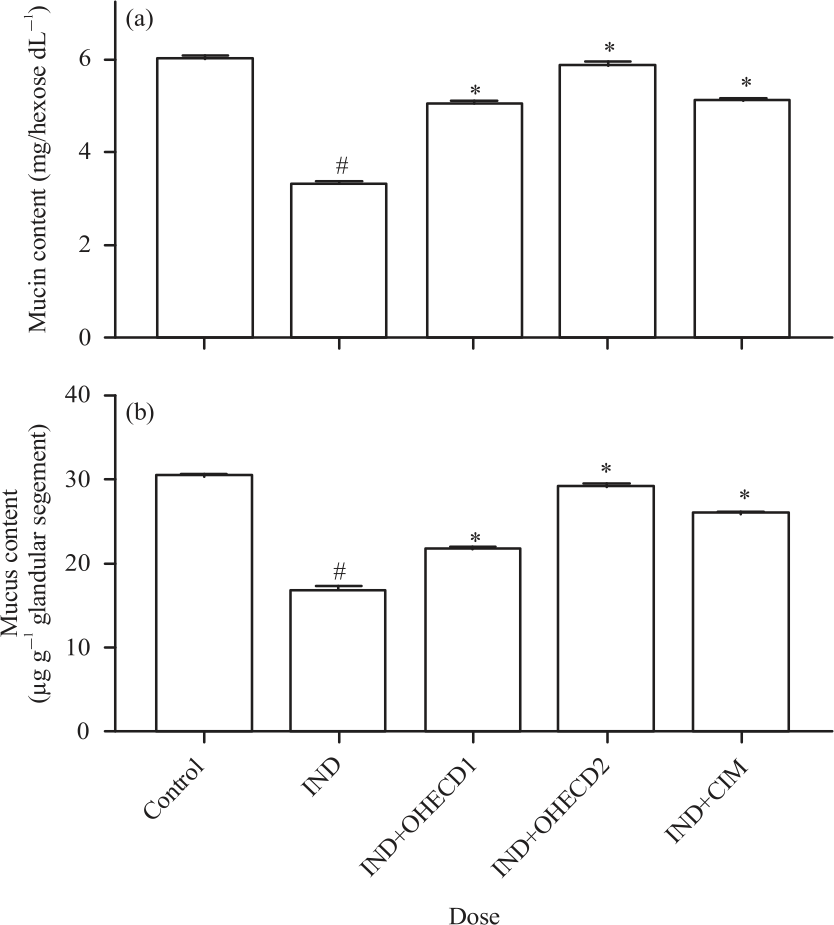
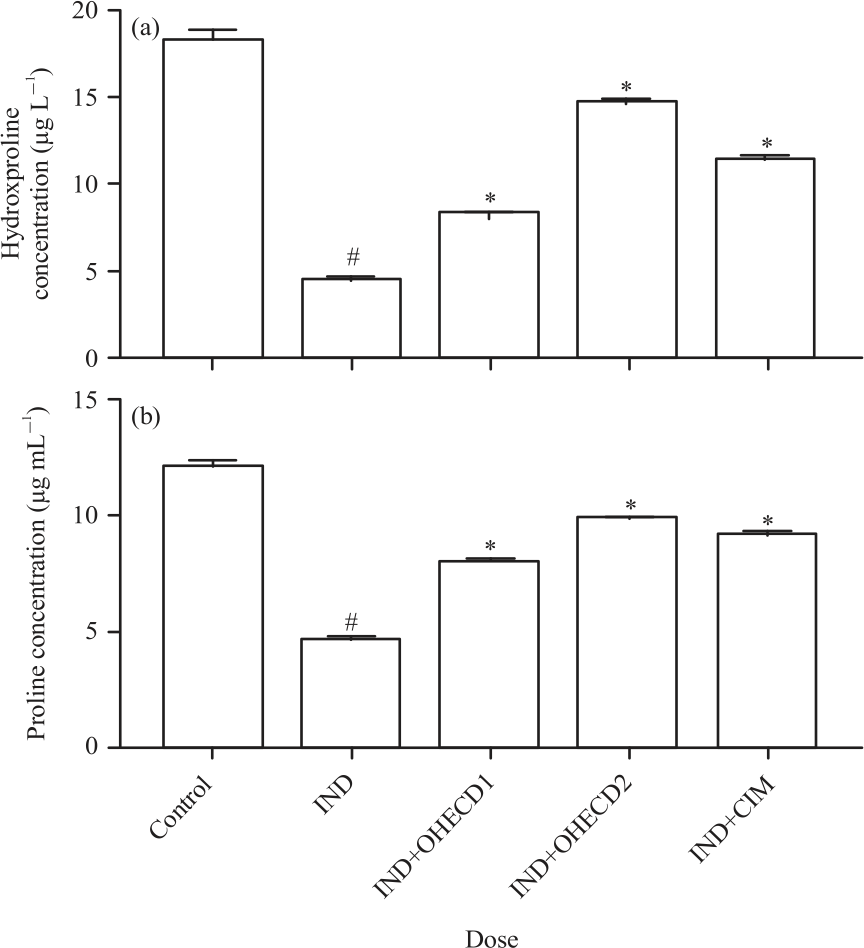
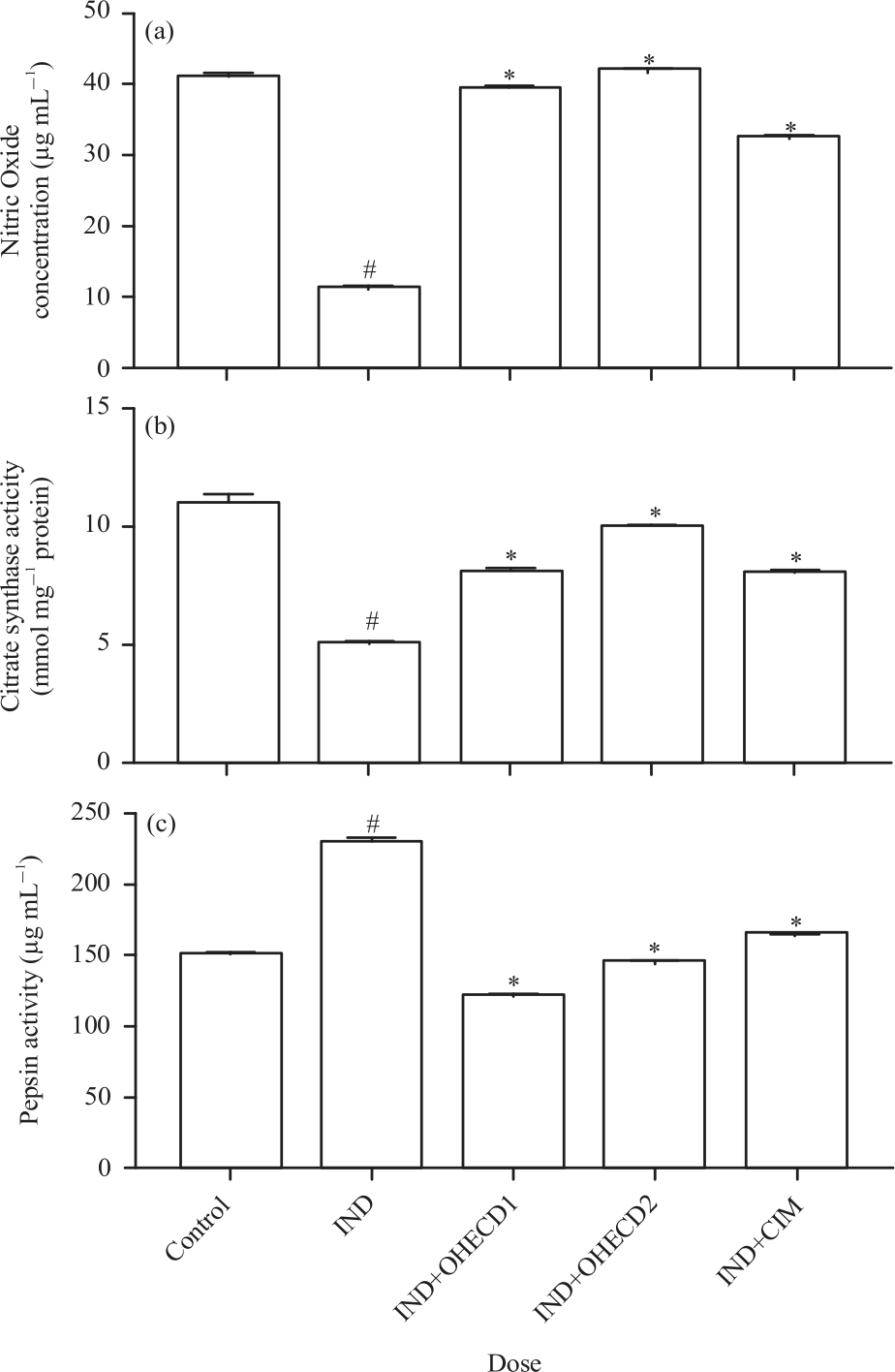
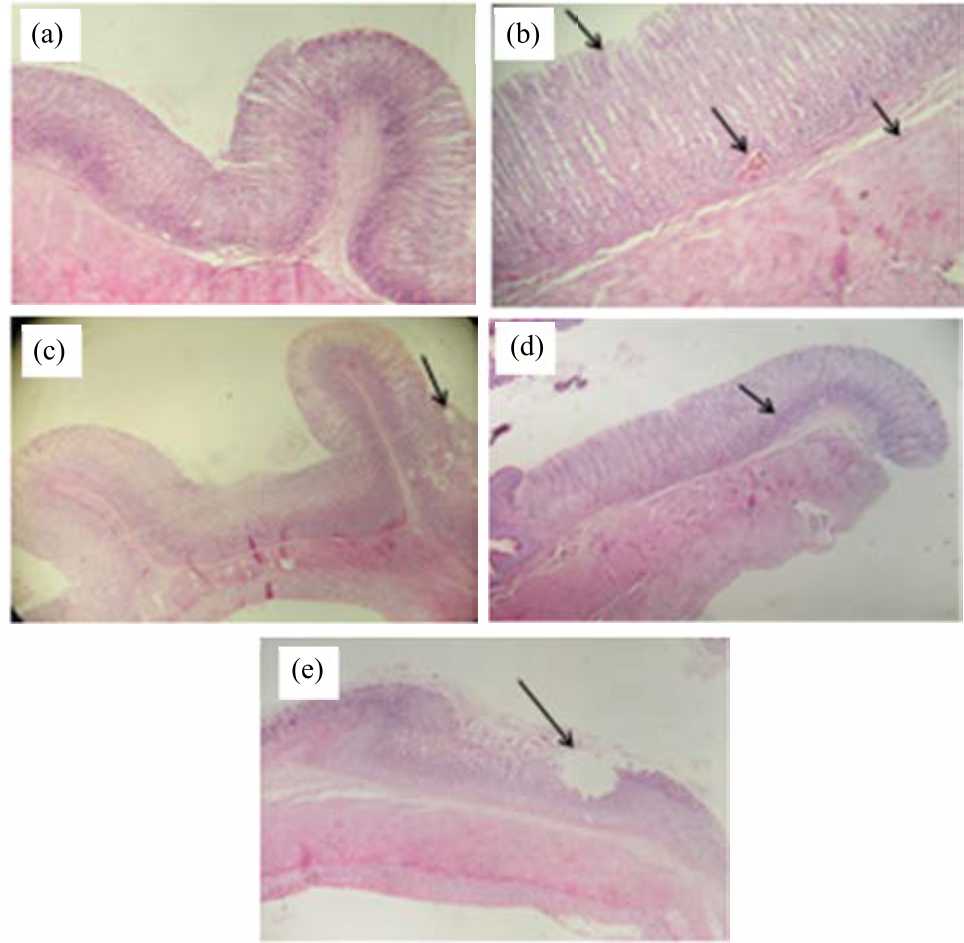
Research Article
Regulation of Connective Components in Indomethacin-induced Gastric Ulcer Healing in Wistar Rats
Department of Biochemistry, Bowen University, Iwo, Osun State, P. O. Box 284, Nigeria
LiveDNA: 234.31317
Fasawe M. Oluwaseun
Department of Biochemistry, Bowen University, Iwo, Osun State, P. O. Box 284, Nigeria
Mrakpor O. Ono-jefe-eroro
Department of Biochemistry, Bowen University, Iwo, Osun State, P. O. Box 284, Nigeria
Ebenezer O. Farombi
Drug Metabolism and Toxicology Research Laboratories, Department of Biochemistry, College of Medicine, University of Ibadan, Nigeria