Research Article
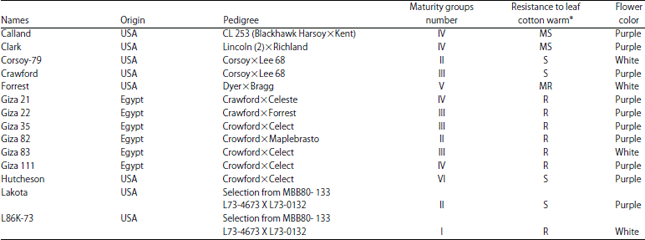
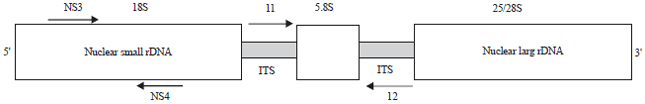
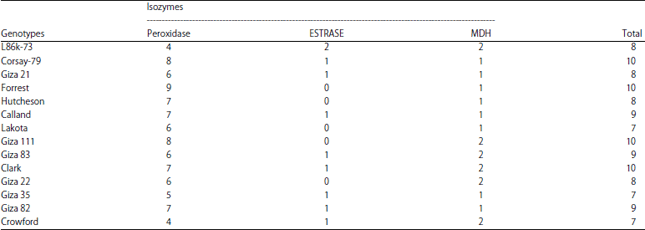
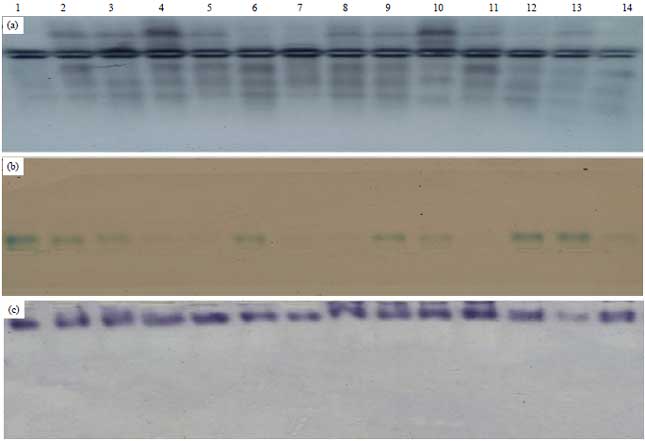
Molecular Characterization of Soybean (Glycine max L. Merr) Genotypes Tolerant and/or Susceptible to Cotton Leaf Worm (Spodoptera littoralis)
Department of Biochemistry, College of Agriculture, Zagazig University, Zagazig 44511, Egypt
LiveDNA: 20.835
Azza Hosni Mohamed
Department of Biochemistry, College of Agriculture, Mansoura University, Mansoura, Egypt
LiveDNA: 20.11836
Mahmoud Imam Nasr
Department of Molecular Biology, Genetic Engineering and Biotechnology Research Institute (GEBRI), University of Sadat, Egypt
Khalil Abdel Hameed El-Halafawy
Department of Molecular Biology, Genetic Engineering and Biotechnology Research Institute (GEBRI), University of Sadat, Egypt
Elsayed Abdel Khalek Alabsawy
Department of Molecular Biology, Genetic Engineering and Biotechnology Research Institute (GEBRI), University of Sadat, Egypt
Ahmed Ismail Hamdi
Department of Food legume Research, Field Crops Research Institute (FCRI), Agricultural Research Center (ARC), Egypt
Ehab Mohamed Zayed
Department of Cell Research, Field Crops Research Institute (FCRI), Agricultural Research Center (ARC), Egypt