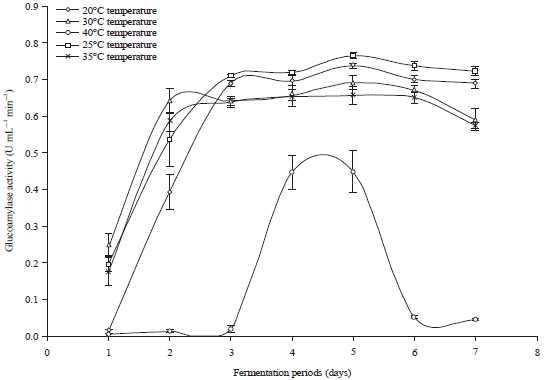
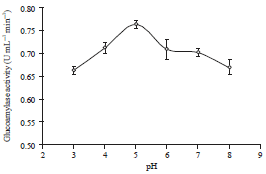
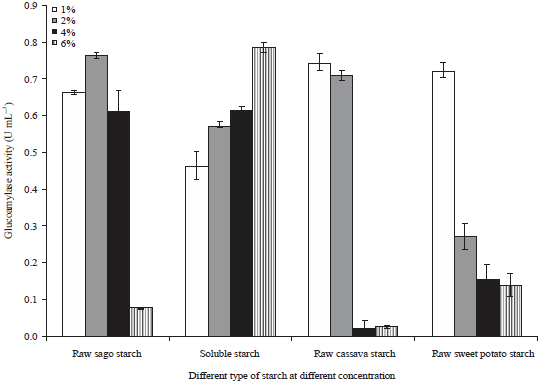
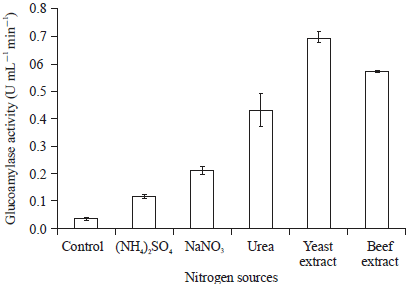
Research Article
Production and Characterization of Crude Glucoamylase from Newly Isolated Aspergillus flavus NSH9 in Liquid Culture
Institute of Nutrition and Food Science, University of Dhaka, Dhaka 1000, Bangladesh
LiveDNA: 880.17997
Ahmad Husaini
Department of Molecular Biology, Faculty of Resource Science and Technology, Universiti Malaysia Sarawak, 94300 Kota Samarahan, Sarawak, Malaysia
Tasmia Tasnim
Department of Nutrition and Food Engineering, Faculty of Allied Health Science, Daffodil International University, Dhaka 1207, Bangladesh