ABSTRACT
The fresh green and yellow banana peel of five different varieties of (Musa, CV, Cavendish) fruits were treated with 70% ethanol, further partitioned with chloroform and ethylacetae (EtoAc) sequentially. The antioxidant activities of the extracts were evaluated by using hydroxyl radical scavenging activity, lipid peroxidation assay, estimation of vitamin C, catalase, peroxidase. The EtoAc extract of type-III and water extract of type-IV had shown the antioxidant activity.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajbmb.2014.122.129
URL: https://scialert.net/abstract/?doi=ajbmb.2014.122.129
INTRODUCTION
There is a great interest in the role of antioxidants in human health which has prompted research in the fields of food sciences and horticulture to assess fruit and vegetable antioxidants (Kalt et al., 1999). Fruits and vegetables contain many different antioxidants components. For example, some flavonoids (including flavones, isoflavones, flavonones, anthocyanins, catechin and isocatechin) that are frequent components in the human diet have demonstrated strong antioxidant activities (Kris-Etherton et al., 2002). Phenolics are the most wide spread secondary metabolites in the plant kingdom. This diverse group of compounds has received much attention as potential natural antioxidants (Sanchez-Moreno et al., 2000). The antioxidant activity of phenolics is mainly because of their redox properties which allow them to act as reducing agents, hydrogen donors, singlet oxygen and metal chelators. Their antioxidant activity is generally based on the number and location of hydroxyl groups present as well as the presence of a 2-3 double bond and 4-oxo function (Karadeniz et al., 2005).
Bananas are one of the most popular fruit carries a number of beneficial pharmacological effectand comes with a set of variety and it is distributed all over the world. It grows in humid low land to upland tropical areas (Banerjee et al., 2010). Being as a tropical plant, banana protects itself from the oxidative stress caused by strong sunshine and high temperature by producing large amounts of antioxidants (Mokbel and Hashinaga, 2005). Active constituents may present in different plant part like fruit, flower, bark, root etc (Banerjee et al., 2010). The medicinal parts used are fruits as well as peels, leaves and the juice from corm. The root is used as an anthelmintic and for reducing bronchocele. The fruit has been used as part of anti-ulcer diet. Anti microbial and antibiotic properties are found in the peel and pulp of fully ripe banana (Ferdinand et al., 2009). Banana peel is an under utilized source of phenolic compounds. It accounts for 40% of the total weight of fresh banana and these are used as fertilizer or discarded in many countries. According to National Cancer Standard Institute banana peel extract is described as a non-toxic to normal human cells, so it can be utilized as a natural source of antioxidants (Lee et al., 2010). It contains various antioxidant compounds such as gallocatectin and dopamine and it is also the rich source of total phenolics and this in turn reflects their antioxidant activity. Several flavonoids have been reported-gallocatechin, catechin and epicatechin. Of these, gallocatechin exhibited the greatest antioxidant activity and was much higher in banana peel (Someya et al., 2002).
MATERIALS AND METHODS
The fresh unripe and fresh ripe bananas were collected from banana growing fields in and around Bangalore. They were screened for their variety at Garden City College, Department of Botany and identified as.
Varieties:| Type I: | Musa cavendisii (AAA) |
| Type II: | Musa pardisia (Diploid) |
| Type III: | Musa acumanate×Musabulbisiania (AAB) |
| Type IV: | Musa bulbisiania×Musa acuminates (ABB) |
| Type V: | Musa sapientum var. Paradisica |
Extraction: The peel tissue of various types of fresh banana fruit (300 g) at green with the trace of yellow stage were selected and was cut into pieces and then heated in 1 L of distilled water for 2 min for the protein coagulation. The peel was homogenized and extracted with 70% acetone, filtered and concentrated to 200 mL.
Followed by partitioned into chloroform (CHCl3) and H20, then extracted with aqueous saturated ethyl acetate. Ethyl acetate, water and chloroform extracts were collected and used for the determination of antioxidants activity.
Antioxidant assay using hydroxyl radical scavenging activity: The method described by Hutadilok-Towatana et al. (2006). Various amounts of test samples were mixed with 134 μL of 30 mM KH2PO4. KOH buffer (pH-7.4) 67 μL of 17 mM deoxyribose, 33 μL of 34 mM H2O2, 33 μL of 1.2 mM EDTA and 67 μL of 300 μM FeCl3. A 67 μL of aliquot of 0.6 mM ascorbic acid was then added to start the reaction. After incubation at 37°C for 1 h, the product of the hydroxyl radical attack on deoxyribose were determined by adding 333 μL of 1%(w/V) TBA (Thiobarbituric acid) in 50 mM NaOH, followed 333 μL of 2.8% (w/v) TCA (Trichloroacetic acid). After further incubation at 80°C for 20 min, the reaction mixtures were centrifuged. The absorption of the clear supernatants was then measured at 532 nm. A parallel assay omitting the test sample acted as a control, where as the normal reaction mixture with outdeoxyribose was used as a sample blank.
Antioxidant assay using lipid peroxidation assay: This assay was done by Liu and Ng (2000) method. The 0.3 mL of the test sample was mixed with 0.1 mL of 10 μM FeSO4 and 0.1 mL of 0.1 mM ascorbic acid at 37°C for 1 h. The reaction was then stopped by addition of 0.75 mL of 28% (w/v) trichloroacetic acid (TCA) and 0.5 mL of 1% thiobarbituric acid (TBA), successively. The mixture was then heated at 100°C for 45 min. After centrifugation, all precipated proteins were removed and the colour was measured at 532 nm. The percent inhibition was calculated.
Estimation of vitamin C, catalase and peroxidase: It was done by the method described by Sadasivam and Manickam (1996).
RESULTS AND DISCUSSION
In the present study, several biochemical constituents and free radical scavenging activities of five varieties of banana were evaluated. Free radicals are involved in many disorders like cancer, AIDS etc. Antioxidants due to their scavenging activity are useful for the management of diseases.
Vitamin C, potential antioxidant having an ability to scavenge a wide range of reactive oxygen species, like peroxide anion, singlet oxygen and hydrogen peroxide and acts as a chain breaking antioxidant. This chain breaking antioxidant property impairs with the formation of free radicals in the process of formation of intracellular substances throughout the body, including collagen, bone matrix and tooth dentine (Beyer, 1994). The quantitative determination of ascorbic acid in plant extracts shows that they are good source of ascorbic acid.
Figure 1 shows the values of vitamin C of the all varieties of banana. From the figure it is clear that concentration of vitamin C is maximum in EtoAc extract of type-III, whereas the other all shown the concentration in a considerable amounts.
Figure 2 shows the values of catalase of the all varities of banana. From the figure it is clear that the concentration of catalase was maximum with water extract of type-IV. Expect type-I (EtoAc) all others have shown very less. Catalase is one of the most important antioxidant enzymes scavenging the active oxygen species in plant cells (Huang et al., 2007). Catalase may be major antioxidant enzyme involved in plant defense mechanism because it is induced during heating and persists during cold storage (Sala and Lafuente, 2000).
Figure 3 shows the values of peroxidase indicated that the maximum activity was found with EtoAc of type-III and the chloroform extract of type-I shown the second next, whereas the other types shown less values.
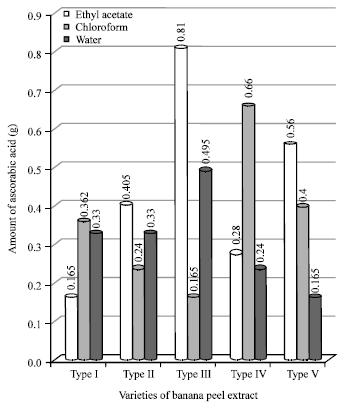 |
| Fig. 1: | Concentration of vitamin C in varieties of banana peel extracts |
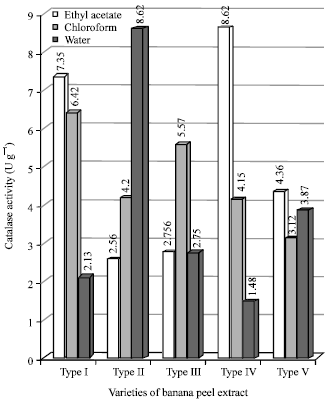 |
| Fig. 2: | Catalase activity in varieties of banana peel extracts |
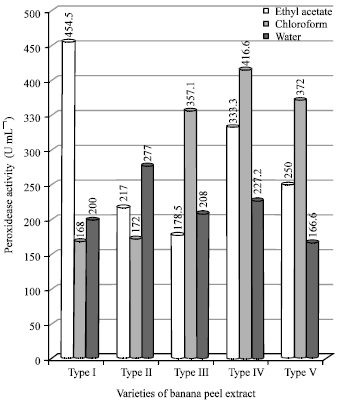 |
| Fig. 3: | Peroxidase activity in varieties of banana peel extracts |
Peroxidase participates in a great number of physiological processes, such as the biosynthesis of lignin and ethylene, defense against pathogens and wounding, auxin metabolism and stress response (Rathod and Yesane, 2011). Peroxidase is recognized to be one of the most heat stable enzymes in plant and its resistance to heat has been reported by a numerous workers (Muftugil, 1985).
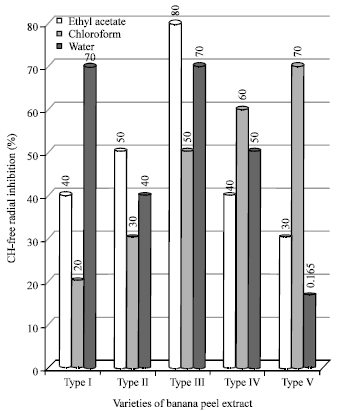 |
| Fig. 4: | Percentage of hydroxyl radical scavenging in varieties of banana peel extracts |
It is reported that peroxidase activity increased when higher temperatures were applied to Phalaenopsis fruits (Ali et al., 2005). The activation of peroxidase is correlated to the defense responses of fruit in presence of pathogens (Muftugil, 1985).
Figure 4 shows the values of hydroxy radical scavenging activity of different varieties of banana. It indicates that maximum was found with ethylacetae extract of type III and chloroform extract of type I and V, where as others II and IV has shown very less activity. The OH radicals have been implicated to play a critical role in the physiological control of cell function (Droge, 2002). The OH radicals react with extremely high rate constants, indiscriminately with almost every type of molecule found in living cells: Sugars, amino acids, phospholipids, DNA bases organic acids and may change normal physiological functions of the cells. Moreover in rheumatic arthritis and related disorders, the reaction of nitric oxide with super oxide generates peroxide nitrite which under acid condition often found in regions of inflammation and ischemia yields hydroxyl radicals (Brown-Galatola and Hall, 1992). The hydroxyl radicals thus generated in the above reaction are believed to contribute to membrane damage of the cells in the region of inflammation. The redox active antioxidants like ascorbate, glutathione, urate, flavonoids, tocpherol, carotenoids andhydroxycinnamic acids present in plants may be the contributing factors for scavenging of OH-radicals (Gacche and Dhole, 2006).
Figure 5 shows the results of lipid peroxidation assay. The data showed that the water extract of type III and type IV were maximum, than the ethyl acetate extract of type III, where as the other type I and type III had showed maximum lipid peroxidation activity. In addition to their free radical scavenging activities, we also evaluated the plant extracts for their abilities to protect biomolecules from oxidative damage, by performing lipid peroxidation assays.
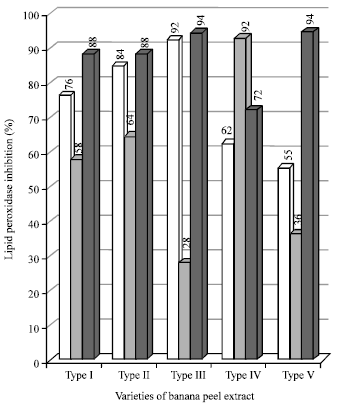 |
| Fig. 5: | Percentage of lipid peroxidation in varieties of banana peel extracts |
Oxidation of unsaturated fatty acids in biological membranes leads to the formation and propagation of lipid radicals, uptake of oxygen, rearrangement of the double bonds in unsaturated lipids and eventual destruction of membrane lipids with production of breakdown products.
CONCLUSION
From the study, it is clear that, among five varieties, ethyl acetate extract of type-III showed the higher concentration of vitamin-C and peroxidase and catalase was maximum in water extract of type-IV. The free radical scavenging activity was found maximum in ethyl acetate extract of type-III and chloroform extract of type-I and type-V. Water extract of type-III and IV has shown the maximum lipid peroxidation activity. From the above, we can conclude as the above tested varieties of banana have the significant antioxidant and free radical scavenging activity. Based on the results we can suggest as the samples can be into considerations for the pharmacological preparations of antioxidants.
We have shown in our present study that banana peel is highly rich in catalase and peroxidase enzymes. Therefore the impact of these antioxidants can be used favorably by man kind by making consciousness effort to leave the inner peel while consuming the bananas. Secondly, the presence of antioxidants in inner peel of banana could be the main source of removing toxic free radicals that may be constantly generated during the growth of banana to maturity. This could be the primordial reason for banana to be left fresh at room temperature even if for more number of days in tropical countries, given that tropical countries are relatively warm than their counterparts which are temperate, the chances of various bacteria and other microorganisms are kept at bay from attacking the banana fruit.
REFERENCES
- Kalt, W., C.F. Forney, A. Martin and R.L. Prior, 1999. Antioxidant capacity, Vitamin C, phenolics, and anthocyanins after fresh storage of small fruits. J. Agric. Food Chem., 47: 4638-4644.
CrossRefPubMedDirect Link - Kris-Etherton, P.M., K.D. Hecker, A. Bonanome, S.M. Coval and A.E. Binkoski et al., 2002. Bioactive compounds in foods: Their role in the prevention of cardiovascular disease and cancer. Am. J. Med., 113: 71-88.
CrossRefPubMedDirect Link - Sanchez-Moreno, C., A. Jimenez-Escrig and F. Saura-Calixto, 2000. Study of low-density lipoprotein oxidizability indexes to measure the antioxidant activity of dietary polyphenols. Nutr. Res., 20: 941-953.
CrossRefDirect Link - Karadeniz, F., H.S. Burdurlu, N. Koca and Y. Soyer, 2005. Antioxidant activity of selected fruits and vegetables grown in Turkey. Turk. J. Agric. For., 29: 297-303.
Direct Link - Mokbel, M.S. and F. Hashinaga 2005. Antibacterial and antioxidant activities of banana (Musa, AAA cv. Cavendish) fruits peel. Am. J. Biochem. Biotechnol., 1: 125-131.
CrossRefDirect Link - Fagbemi, J.F., E. Ugoji, T. Adenipekun and O. Adelowotan, 2009. Evaluation of the antimicrobial properties of unripe banana (Musa sapientum L.), lemon grass (Cymbopogon citratus S.) and turmeric (Curcuma longa L.) on pathogens. Afr. J. Biotechnol., 8: 1176-1182.
Direct Link - Lee, E.H., H.J. Yeom, M.S. Ha and D.H. Bae, 2010. Development of banana peel jelly and its antioxidant and textural properties. Food Sci. Biotechnol., 19: 449-455.
CrossRefDirect Link - Someya, S., Y. Yoshiki and K. Okubo, 2002. Antioxidant compounds from bananas (Musa cavendish). Food Chem., 79: 351-354.
CrossRefDirect Link - Hutadilok-Towatana, N., P. Chaiyamutti, K. Panthong, W. Mahabusarakam and V. Rukachaisirikul, 2006. Antioxidative and free radical scavenging activities of some plants used in Thai folk medicine. Pharm. Biol., 44: 221-228.
CrossRefDirect Link - Liu, F. and T.B. Ng, 2000. Antioxidative and free radical scavenging activities of selected medicinal herbs. Life Sci., 66: 725-735.
CrossRefDirect Link - Beyer, R.E., 1994. The role of ascorbate in antioxidant protection of biomembranes: Interaction with vitamin E and coenzyme Q. J. Bioenergetic Biomembr., 26: 349-358.
CrossRefPubMedDirect Link - Huang, R., R. Xia, L. Hu, Y. Lu and M. Wang, 2007. Antioxidant activity and oxygen-scavenging system in orange pulp during fruit ripening and maturation. Sci. Hortic., 113: 166-172.
CrossRefDirect Link - Sala, J.M. and M.T. Lafuente, 2000. Catalase enzyme activity is related to tolerance of mandarin fruits to chilling. Postharvest Biol. Technol., 20: 81-89.
CrossRefDirect Link - Rathod, S.V. and D.P. Yesane, 2011. Isolation of antioxidant enzymes from some wild edible fruits at mature and ripened stage rhizome. Curr. Bot., 2: 53-55.
Direct Link - Muftugil, N., 1985. The peroxidase enzyme activity of some vegetables and its resistance to heat. J. Sci. Food Agric., 36: 877-880.
CrossRefDirect Link - Ali, M.B., E.J. Hahn and K.Y. Paek, 2005. Effects of light intensities on antioxidant enzymes and malondildehyde content during short-term acclimatization on micropropagated Phalaenopsis plantlet. Environ. Exp. Bot., 54: 109-120.
CrossRefDirect Link - Droge, W., 2002. Free radicals in the physiological control of cell function. Phsyol. Rev., 82: 47-95.
CrossRefPubMedDirect Link - Brown-Galatola, C.H. and N.D. Hall, 1992. Impaired suppressor cell activity due to surface sulphydryl oxidation in rheumatoid arthritis. Rheumatology, 31: 599-603.
CrossRefDirect Link - Gacche, R.N. and N.A. Dhole, 2006. Antioxidant and possible anti-inflammatory potential of selected medicinal plants prescribed in the indian traditional system of medicine. J. Pharma. Biol., 44: 389-395.
CrossRefDirect Link








