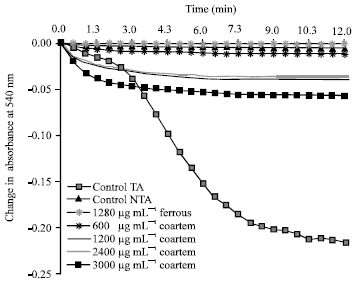
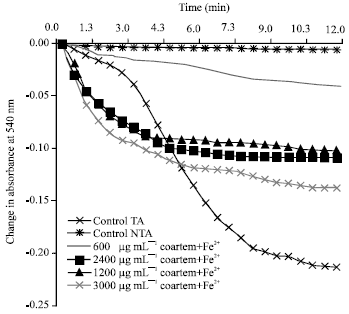
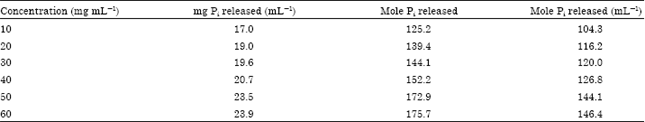
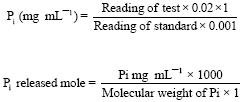Research Article
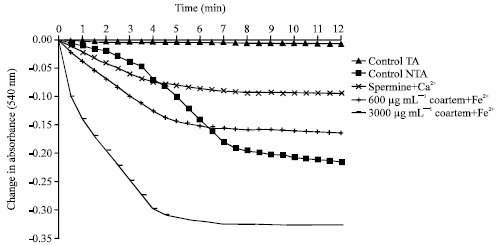
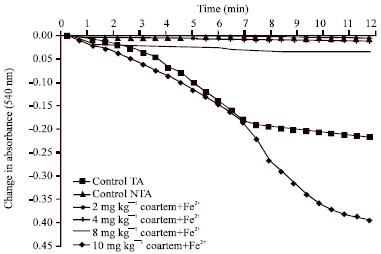
Induction and Uncoupling of Rat Liver Mitochondria by Oral Administered Coartemether
Department of Biochemistry, Ben Carson School of Medicine, College of Health and Medical Sciences, Babcock University, Ilisan-Remo, Ogun State, P.M.B. 21244, Ikeja, Nigeria
LiveDNA: 234.33615
Oluwadamilola M. Odunaike
Department of Biochemistry, Ben Carson School of Medicine, College of Health and Medical Sciences, Babcock University, Ilisan-Remo, Ogun State, P.M.B. 21244, Ikeja, Nigeria
Olufunso O. Olorunsogo
Biomembrane and Biotechnology Laboratories, Department of Biochemistry, Faculty of Basic Medical Sciences, College of Medicine, University of Ibadan, Ibadan, Nigeria