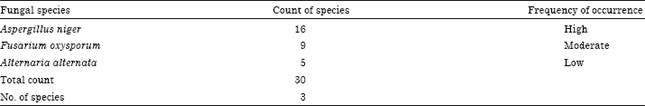
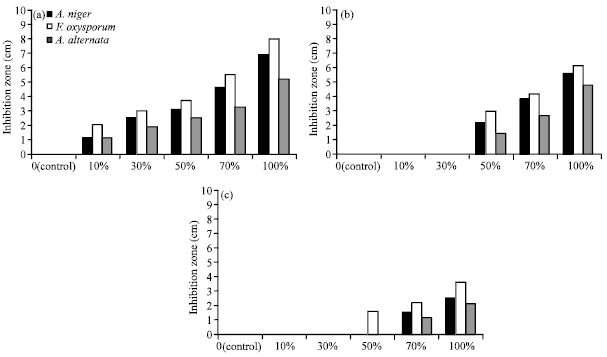
Research Article
Comparative Efficacy of Some Plant Extracts against Fungal Deterioration of Stucco Ornaments in the Mihrab of Mostafa Pasha Ribate, Cairo, Egypt
Department of Conservation and Restoration, Faculty of Archaeology, Cairo University, Giza 12613, Egypt
LiveDNA: 20.28143