ABSTRACT
The Oil palm is as old as creation. Every part of the tree is useful economically and for domestic purposes. The oil Palm (Elaeis guineesis jacq.) is believed and accepted to have originated from West Africa. In Nigeria, it is cultivated in the South East Zone and the Niger Delta areas. The mesocarp of its fruit yields Palm oil which is orange-red in colour, due to the presence of the carotenoids. The kernel contains the second oil called the Palm Kernel Oil (PKO). The major and minor components of the palm oil play numerous health functions in humans. Some metabolites which play notable roles in the biosynthesis of triglycerides and products of lipolytic activities have been detected in the palm oil. Arising from the wide array of the components of palm oil, researches have been conducted which involved studies with the humans and animals. Results obtained from the researches have been of immense contribution to human health and products that could be developed from the palm oil. Additionally, the palm yields a nutritious sap when tapped. The components of the palm sap play significant roles in human physiology. This review paper is set to produce an update on some aspects of the oil palm, the products and their implications in human health.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajbmb.2012.106.119
URL: https://scialert.net/abstract/?doi=ajbmb.2012.106.119
INTRODUCTION
The Elaeis guineensis jacq. is the common African oil palm. The stem is stout and stands erect. It could attain a height of 30 m when fully grown. The plant is monoecious (bears both the male and female flowers). When four spear leaves appears on the palm it is matured for tapping. Tapping the base of the spear leaf produces exudates called the palm sap or palm wine. The fruit is a drupe. The fruit produces two types of oil; the palm oil from the mesocarp and the kernel oil from its kernel. The crude palm oil is orange-red in colour obtained by milling mechanically or by using the traditional method (Hartley, 1977). Red palm oil is the main source of domestic or edible oil in Africa. The crude palm oil could be fractionated and refined into products like Palm Olein and Super Olein. The fractionation brings about an increase in monounsaturated oleic acid with the concomitant reduction of palmitic acid, the major saturated fatty acid (Edem, 2002). Red palm oil contain 50% saturated, 40% monounsaturated, 10% polyunsaturated fatty acids and it is used for combating vitamin A deficiency in developing countries (Rukmini, 1994). Several nutrients which play significant roles in human metabolic processes and growth have been identified in red palm oil. For example, the vitamin E Content had been reported (Chong and Ng, 1991; Sundram and Top, 1994; Goh et al., 1985). The carotenoids give the palm oil its characteristic colour (Cottrell, 1991). The carotenoids together with vitamin E, ascorbic acid, enzymes and proteins are members of the biological antioxidant network converting highly reactive radical (*OH) and free fatty proxy radials to less active species, thus protecting against oxidative damage of cells. The vitamin A content of the red palm oil plays important roles in growth, development and in visual process (Ndon, 2006a; Edem, 2009).
The carotene can be removed either by bleaching using suitable chemicals or by heating the oil to high temperature (above 1800°C). The use of palm oil for frying purposes at high temperatures lead to losses of the beta-carotenes and the vitamin C contents as reported by Inocent et al. (2011) and Abong et al. (2011). Palm oil usually contains various percentages of Free Fatty Acids (FFA) as a result of fat splitting reactions in which glycerin molecule combines with water to yield FFA. Drying of palm oil reduces the amount of FFA during storage. The effect of light bearing different wavelengths on Free Fatty Acids (FFA) of stored crude palm oil has been reported. Results indicated that the FFA of the oils increased with storage time. The FFA values of the oil did not follow any established order, especially as related to the spectrum of light (Henry, 2011). Palm oil has minor constituents such as sterols (0.03%), phosphatides (0.01%) and tocopherols (0.05%) (Ndon, 2006a).
The major carotenoids found in palm oil are the beta-carotene. The carotenoids in red palm oil are the most important minor constituent. When the carotene molecule is splited by the addition of the elements of water to both halves, vitamin A is produced. In beta-carotene the two halves are identical but in the other carotenes they are not, only one-half being of the constitution is necessary for vitamin A formation (Hartley, 1977).The types of carotenes in Palm oil Mill Effluent (POME) have been identified by Ahmad et al. (2010), by making use of adsorption chromatography.
Phospholipids forms part of the minor constituents of the palm oil. The mesocarp oil of Elaeis guineensis provides 1000-2000 ppm of phospholipids (Goh et al., 1983).
The use of palm oil in preservation purposes has been reported. Law-Ogbomo and Egharevba (2006) used the palm oil along with other vegetable oils in the control of cowpea weevil, Callosobruchus maculatus. The effect of palm oil in protecting stored grains from Sitophilus zeamais and Callosobruchus maculatus was reported by Abulude et al. (2007).
The Palm oil Mill Effluent (POME) is a useful source for the generation of biohydrogen and production of butyrate, propionate and acetate as demonstrated by Yusoff et al. (2009).
This review is set to examine the origin, distribution and propagation of the oil palm. Additionally, to examine the characteristics, chemical, biochemical, nutritional and the health implications of the minor and major components of the palm oil.
ORIGIN
It is globally accepted that the oil palm originated from West Africa. The natural habitat of the oil palm is in fresh water forest of West Africa. The oil palm has long been domesticated by many West African coastal dwellers before the advent of the white man to West Africa. In recent years, fossil records have also supported the West African origin for palm oil. For example, there is evidence that palm oil was being used in Ancient Egypt in 3000 BC based on a jar of palm oil excavated from a tomb in Abydos. The oil palm was believed to have been taken to Egypt by Arab traders (Ndon, 2006a). Corley and Teo (1976), reported on the origin of the oil palm and claimed that this tree crop from West Africa was first introduced to Brazil and other tropical countries in the 15th century by the Portuguese but the propagation did not start immediately until the 19th century.
Authors, like Hartley (1988) have also contributed to the origin of the oil palm. The former claimed that oil palm originated from West Africa but the latter reported that there is considerable linguistic evidence that the Brazilian terms describing oil palm are of Africa origin.
Although, the oil palm originated from West Africa, South-East Asia, particularly Malaysia and Indonesia, are now the leading producers of palm oil and palm kernel (Ndon, 2006a).
DISTRIBUTION IN AFRICA
Ndon (2006a), Hartley (2000) and Agboola (1979) have adequately reported on the distribution of oil palm in Nigeria. Under the Nigerian context, three major areas have been identified and classified for oil palm distribution. They include; The dense oil palm gloves; the oil palm bush and the secondary rain forest with oil palms.
Oil palm distribution in Africa has also been reported by Seven (1967). The oil palm could spread widely where the climate was acceptable and there was human farming activity. Some movements of seed may have been due to other species but humans are clearly by far the most factors in its distribution.
In Nigeria, there are no semi-wide Palms north of about 7°N, except in particularly favoured areas where there are shallow water tables, such as near the Jos Plateau (Corley and Tinker, 2003).
The Palm belt of Africa runs through Sierra Leone, Liberia, the Ivory Coast, Ghana, Togol and Benin (previously Dahomey), Nigeria, the Cameroons, the Peoples Republic of Congo (formerly Zaire).
CULTIVATION: SOIL AND CLIMATE
The oil palm has been grown successfully in about 20 countries which lie between 10°N and S of the equator, in a wide range of tropical climate and soils. To achieve maximum yield, a mean maximum temperature of 29-33°C and a minimum temperature of 22-24°C has been recommended (Ndon, 2006a). Low or cold temperatures reduces the oil palm growth, prolong fruiting and ripening of the fruits. These include, increased abortio and slow vegetative growth and bunch ripening. Hartley (1988), reported the effects of the cultivation of oil palm in an area of high altitude, 500 m and low temperature which indicated that palms came into bearing at least a year later than palms grown at usual a ltitude. Chapman et al. (2001) reported the breeding of oil palms in an area of high and low temperature, though there seems to be little demonstrable progress, a commercial plantation has been emarked for Ethiopia in high locations (Corley and Tinker, 2003).
The rainfall requirement for the oil palm is a total of 2000 mm or more per year to maintain a continuous supply of soil moisture content. The even distribution of rainfall throughout the year is considered adequate for the successful cultivation of the oil palm (Hartley, 1988; Ndon, 2006a). Rainfall is the most critical determinant of the climatic factors in the oil palm cultivation. The water relations and palm yield have been investigated and well documented (Foong, 1993; Kee, 1995; Tinker and Nye, 2000; Hartley, 1988; Ochs and Daniel, 1976; Caliman and Southworth, 1998; Ndon, 2006a).
Soil and climate are the basic resources for growing crops. It is possible to amend either in some ways such as the chemical composition of soil with fertilizers or the rainfall regime with irrigation of rain saving systems but such amendments can be expensive. Other components of these resources, such as the physical nature of soils or the temperature regime in climate, are virtually impossible to alter on a large scale.The oil palm requires high temperature, being an equatorial tree crop. The temperature range of 24-28°C has been considered ideal for its growth, though it is difficult to separate the effects of maximum and minimum temperature (Corley and Tinker, 2003). Prolonged drought stress, increases the number of unopened leaves, bunch desiccation, blast diseases, abortion, reduces sex ratio and oil palm yield. The use of irrigation, mulching of the soil with EFB and pruned fronds and conservation of moisture improves yield of drought affected palms (Ndon, 2006a). A search for such relationship between the water deficit and palm growth and yields can be made using statistical models (Corley and Tinker, 2003). Earlier work of Hartley (1988), using the IRHO method, revealed that an additional water deficit of 100 mm in a year of harvest decreased yield by 10%. The findings of Ochs and Daniel (1976) indicate that a 100 mm water deficit resulted in the reduction of FFB yield between 10 and 20%, influenced by the soil quality. Additionally, Caliman and Southworth (1998), concluded in their findings that a deficit of 100 mm in a particular year resulted in FFB yield reduction of 8-10% in the first following year and by 3-4% in the second year.
Authors like Hartley (1988), Corley and Tinker (2003) and Ndon (2006b) have reported on the different soils from the areas of the world, where the oil palm is grown; e.g., soils of Asia, Indonesia, Africa, democratic Republic of Congo and America. The different soils used for the cultivation of the oil palm in Mexico have been reported by Wilson-Bibiano et al. (2001). The soils which support the growth of the oil palm both wild and planted have been reported as ‘Acid sands’ soils. Most of the palms in the Nigerian palm belt grow largely on unconsolidated sandstones or ‘Benin Sand’ which were classified as ‘fascs’ by Vine (1956); Tinker and Ziboh (1959). Ogunkunle et al. (1999) however, redefined these soils.
The Malaysian soil which supports the growth of the oil palm is either the ‘inland’ soils or ‘coastal’ soil (Corley and Tinker, 2003).
Oil palm grows in a wide range of tropical soils, provided that there is adequate supply of water. The topography and slope, soil moisture supply, soil physical and chemical properties are important parameters which supports the growth of oil palm. Oil palm is best planted on flat land or slopes of less than 23% (<12°). However, soil physical properties are more important than the soil chemical properties because deficient nutrients can be supplied by the addition of mineral fertilizers to improve soil chemical properties (Ndon, 2006b).
PROPAGATION BY TISSUE CULTURE
Existing research findings have indicated that in many perennial crops genetically uniform material can be produced by vegetative propagation, using suckers, cuttings or grafts, where the genetic constitution of all ‘offspring’ is identical with that of the original material. All individual plants with the same genetic composition or constitution, produced from the original material in such ways, resulting in a clone (Corley and Tinker, 2003). Clones cannot be produced by the common technique referred to above, in the case of oil palm, because it has a single growing point and does not suck like other palms in the same family but elite palms can be multiplied through tissue culture technique (Corley and Tinker, 2003; Ndon, 2006b), where clones can be produced, by which small pieces of tissues (explants are grown on special nutrient mediums or solutions, growth regulators including auxins and cytokinins, under controlled light and atmosphere. The tissue gets disorganized and broken down into cells which further develop into calli and finally into plantlets having roots and shoots (Ndon, 2006b).
Research into the possibilities of propagating the oil palm though tissue culture started in the 1960’s and by the mid-1970’s remarkable breakthroughs were reported (Jones, 1974). The earliest workers after the breakthrough into the research of the clonal palms include (Corley et al., 1979; Wooi, 1990; Le Guen et al., 1991). However, major setbacks were observed by the application of this method of propagation which involved abnormal flowering and severe bunch failure (Corley et al., 1986). Research is still underway on the abnormal flowering, eventhough the propagation of the oil palm has been on the increase through tissue culture method.
The advantages and disadvantages of tissue culture technique in oil palm propagation have been reported, to include; uniformity in flowering and fruit set; increase in bunch yields; less variability in bunch composition compared to seedling progenies; improvement in breeding programme and abnormalities; disease and pests problem; high cost. Additionally, the success of clonal propagation will depend on the identification of clones with sufficiently high yields to justify the additional cost of tissue culture, compared with production of seed and perhaps of screening to eliminate abnormalities (Corley and Tinker, 2003). An early estimate of the cost of propagation by tissue culture gave a cost per plant over five times that of a seedling (Corley and Stratford, 1998). The estimated cost of a ramet was put at five times the cost of a seed (Rival, 2000). The presentation of this figure in a financial model indicated that a yield increase of 20% would be needed if clonal planting were to be profitable (Corley et al., 1988). Profitability therefore, would depend heavily on the palm oil price and also on the financial discounts rate but this gives an indication of what the target should be in clone development (Corley and Tinker, 2003).
PROPAGATION THROUGH SEED/KERNEL
Apart from the use of tissue culture technique, the oil palm propagation could be done directly from the seed or nut called the oil palm kernel. Historically, this method has been before the advent of tissue culture technique and is still largely in use by large medium and small scale farmers today. The handling of dormancy of oil palm nuts, factors that induces germination of oil palm nuts, have been reported (Ndon, 2006b). Fresh oil palm seed or nut is always dormant and slow to germinate except certain specific temperature, oxygen and moisture requirements are met. Seed dormancy in the oil palm is caused by two physical barriers that is the hard shell (endocarp) and operculum. If these physical barriers are removed, the oil palm seed germinates easily (Ndon, 2006b).
PHYSICAL, CHEMICAL AND BIOCHEMICAL PROPERTIES OF PALM OIL
Palm oil is liquid which separates into two phases (liquid and solid portions) when allowed to rest. It is extracted from the mesocarp of the freshly orange-red fruit of the oil palm (Elaeis guineensis). It is a lipid which is only soluble in fat solvents and not soluble in water (Corley and Tinker, 2003; Edem, 2002). Pure palm oil is orange-red in colour, depending on the amount of carotenoids present.
The palm oil is made up of both major and minor components (Table 1, 2). Triglycerides form the major components of palm oil, with smaller proportions of diglycerides and monoglycerides while the minor components include, the free fatty acids and phytonutrients. The phytonutrients include: Carotenes, vitamin E (Tocopherol and tocotrienoids), Phytosterols, phospholipids, squalene, phenolic acids, flavonoids, co-enzyme Q10; chlorophyll, phosphatides, alcohols and sterols (Sambanthamurthi et al., 2006).
Phytonutrients can be obtained as by-products from palm oil mill and refinery (Sambanthamurthi et al., 2006) and as by products, during the process of producing biodiesel from crude palm oil (Choo et al., 2002).
| Table 1: | Palm oil components |
 | |
Source: Malaysian palm oil board and International medical university, Malaysia | |
| Table 2: | Minor components of palm oil |
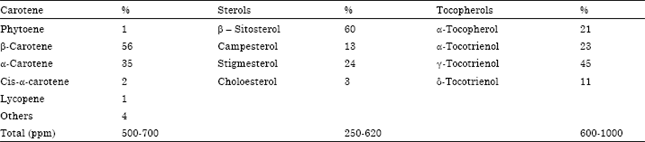 | |
Source: Jalani and Rajanaidu (2000) | |
| Table 3: | Composition of fatty acids in palm oil |
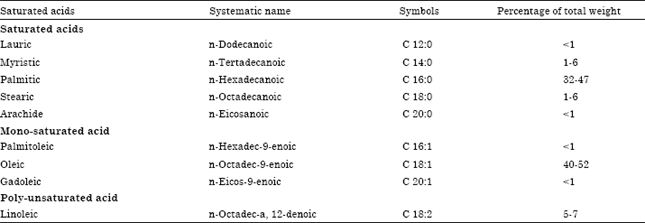 | |
Source: Hilditich and Williams (1964) | |
Additionally, phytonutrients could be obtained as by products of palm fatty acids distillate. Nutritionally, the most important of the minor components of palms oil are the α and β carotenes which are precursors of Vitamin A, tocopherols and tocotrienoid vitamin E (Corley and Tinker, 2003). Amongst the carotenes, β carotene has the strongest pro vitamin A activity.
Triglycerides: Palm oil like vegetable oils and fat is made-up of triglycerides which forms its major constituents. The densities of triglycerides are lower than that of water. They exist as liquid or solid at normal temperature. If solid, they are called ‘fats’or ‘butters’ and ‘oil’when liquid. Triglycerides are made from a molecule of glycerol and three fatty acids. Glycerol being a trihydric alcohol (Containing three-OH hydroxyl groups) can react with three fatty acids to form mono glycerides, diglycerides and triglycerides.
Fatty acids: The fatty acids composition of palm oil is 3-5%. The fatty acids are long hydrocarbon chains that carry a terminal carbonyl group which characterized them as weak acid. The physical nature of the fat is determined by the chain lengths of the fatty acids and by whether, the acids are saturated or unsaturated (Ndon, 2006b).
The Saturated and unsaturated fatty acids of palm oil are stated in Table 3. The Palmitic (32-47%), Oleic (40-52%) and Linoleic (5-7%) are the main fatty acids components of palm oil. The fatty acids contribute about 95% of the total weight of a triglyceride mole and they are the reactive portion of the molecule. Hence, the proportion and composition of these fatty acids affect the physical and chemical properties of palm oil (Ndon, 2006b).
DIET AND HEALTH ASPECTS OF PALM OIL
Researchers have paved way into the understanding of the total composition of the palm oil and thus the significance of palm oil in health. The minor and major components of the palm oil are useful in this regard. The processing and storage methods are equally significant in the preservation of the useful components for health. Though, oils and fats are an essential part of our diet, the benefits gained may depend in part on the composition of the oil (Corley and Tinker, 2003). WHO and other bodies have recommended at least 12 kg per head as annual consumption of oils and fats, but that the oil and fats should not exceed 30% of the daily calorie intake (Corley and Tinker, 2003).
The relationship between diet and the incidence of coronary heart diseases have been studied by several researchers and reported (Hayes and Khosla, 1992; McNamara, 1992; Cottrell, 1991). High fat intake has been linked to increased risk of heart disease. High blood cholesterol level has been associated with increased causes of heart disease. Studies on Cardio Vascular Disease (CDV), one of the leading causes of death in many countries, have focused on ways of reducing plasma cholesterol by nutritional means (Edem, 2002; Onyeneke and Alumanah, 1991). Though, palm oil contains almost equal proportions of saturated and unsaturated fatty acids, the saturated fatty acids, appear to be more harmful than the unsaturated fatty acids. An increase in saturated fatty acids in the diet increases blood cholesterol (Cottrell, 1991). Researchers have also revealed that myristic acid (C14) is more harmful than palmitic and that palmitic is only harmful if the level of linoleic acid in the diet is very low. Palmitic acid may also have undesirable effects if the diet is high in cholesterol but not if it is low (Hayes and Khosla, 1992). The tocotrienols (part of the phytonutrients) in particular the gamma and delta isomers have been shown as capable of lowering and inhibiting cholesterol synthesis at the serum/plasma or at the liver. Additionally, the daily consumption of tocotrienols enriched fraction of palm oil (200 mg Palmvitee capsule) can result in a significant reduction of serum cholesterol, LDL Cholesterol, APOB, thromboxame, platelet factor 4 and glucose of hypercholesterolemic subjects within four weeks of administration (Qureshi et al., 1991; Packer, 1992; Song and DeBose-Boyd, 2006). Diet high in polyunsaturated fats results in lower total cholesterol but HDL-C is reduced, as well as LNL-C. In animal studies poly-unsaturated fats have been associated with increased risk of cancer but this does not appear to have been confirmed in human studies (Corley and Tinker, 2003). Recent studies have also shown that trans-acids tend to increase total cholesterol and to reduce HDL-C and increase LDL-C levels (Sambanthamurthi et al., 2006; Enig, 1998). Past reports have indicated that monounsaturated fatty acids were regarded as neutral but recent studies have shown that oleic acid is as effective as polyunsaturated in lowering total cholesterol and imporving the LDL-C: HDL-C ratio (Sambanthamurthi et al., 2006).
Phytonutrients which have antioxidant properties e.g., tocopherols (vitamin E), Carotenoids (vitamin A) and phospholipids have influence on the quality of the palm oil and also play notable roles in human diet. The roles of these phytonutrients in human health are beginning to attract research interest in palm oil studies, in addition to the mechanism of action of their isomers. Research has revealed that about 600 natural carotenoids exist. Amongst these, 13 have been identified in the crude palm oil. Out of the known 600 carotenoids, 10% have pro-vitamin A activity. Carotenoids are soluble in fats solvents and also require fat for its conversion into vitamin A. The crude palm oil provides a suitable environment for the above action. Several research reports have indicated that the red palm oil is a positive source for the treatment of vitamin A deficiency (Roo, 2000). Additionally, some workers have advised that nursing mothers should take red palm oil as supplement with their food in order to prevent vitamin A deficiency (Lietz et al., 2000). The carotenoids together with vitamin E, ascorbic acid, enzyme and proteins are members of the biological antioxidant network converting highly reactive radicals (*OH) and free fatty peroxy radicals to less active species, thus protecting against oxidative damage in cells (Krinsky, 1992; Hendrich et al., 1994; Packer, 1992). The red palm oil provides high energy density in the food and a rich source of beta carotene which can be split into two vitamin A molecules (Rukmini, 1994). The vitamin A molecules produced, plays significant roles in visual processes, differentiation of cellular epithelium and genetic regulation(Edem, 2002). Vitamin has been known to play great role in normal development and growth in humans (Levin and Davis, 1997).
Phospholipids forms part of the phytonutrients in red palm oil and the main building blocks in all living cells (including human), besides its roles as essential components of lipoproteins. It functions as antioxidant synergists in the presence of vitamin E and other cascade of antioxidants. A list of the main phospholipids in palm oil includes the following, phosphatidylcholine, Phosphatidylinositol, Phosphatidylethanolamine and Phosphatidyl-glycerol.
Supplementation of phosholipids (Choline) has been advocated by researchers to aid in brain development of the growing fetus and lactating mother (Zeisel et al., 1986). Additionally, the administration of phosphatidylserine to improve memory and other brain functions which tend to decline with age has been advocated (Jager et al., 2007). Choline supplementation has also been recommended for athletes involved in energy endurance, as research has revealed that those involved in such exercises are prone to loss in the choline concentration of the blood and hence, replacement is needed at the plasma level. In addition, phosphatidylserine supplementation has been recommended for athletes to promote a desired hormonal status by blunting the increase of cortisol levels (Starks et al., 2008). Phospholipid fractions and phosphatidylcholine are able to ease digestion, nutrient absorption and lipid transportation (Lochmann and Brown, 1997).
Researches into vitamin E (tocopherols and tocotrienols), have revealed their implications in human health. Their benefits in human health include:
Anti-cancer effect: Evidences of the anti-cancer effects of vitamin E (tocopherols and tocptrienols) and their isomers in palm oil have been reported in a comparative study of the effects of tocotrienols and alpha-tocopherols on breast cancer. Results indicated that the tocotrienols fractions were able to induce an inhibitory action on the human breast cancer cells, whereas the alpha-tocopherols were not able (Nesaretnam et al., 2004, 2008; McIntyre et al., 2000).
Antioxidant effects: The tocopherols and tocotrienol of palm oil are known to provide a wide array of antioxidants effects in physiological systems. They are capable of scavenging free radicals or reactive oxygen species, thus protecting the cells against oxidative damage. Alpha tocopherols and gamma tocotrienols have antioxidative effects on lipid peroxidation, in the presence of a xenobiotic metabolizing enzyme that induces lipid peroxiadation (Zuzana et al., 2005). Lipid peroxidation is a chain reaction that provides a continuous supply of free radicals which results in not only food rancidity but also damage to tissues in vivo, leading to cancer, inflammatory diseases, atherosclerosis and ageing.
Lowering and inhibition of cholesterol synthesis: The tocotrienols, in particular the gamma and delta isomers have been shown as capable of lowering and inhibiting cholesterol synthesis at the serum/plasma or at the liver. Additionally, the daily consumption of tocotrienols-enriched fraction of palm oil (200 mg palmvitee capsule) can result in a significant reduction of serum cholesterol, LDL cholesterol, APOB, thromboxame, platelet factor 4 and glucose of hyper cholesterolemic subjects within four weeks of administration (Qureshi et al., 1991; Packer, 1992; Song and DeBose-Boyd, 2006).
Cardio-protective ability: Animal trials have shown that the administration of Tocotrienol-rich Fraction (TRF) has resulted in cardio-protective ability (Das et al., 2008). The cardio-protective effects produced by the isomers of tocotrienol was of the order of: γ>α>δ. The inhibition of normal cellular gene, C-Src activation and proteosome stabilization were found to be reasons behind the cardio-protective properties of TRF.
Support to diabetes: Administration of tocopherol, Vitamin E or tocotrienol-rich fractions, have been shown to recover glycemic status, inhibit oxidative damage; prevent DNA damage in animal studies and prevent glycosylation of end-products in serum and decrease in diabetic rats (Budin et al., 2006; Nazaimoon and Khalid, 2002).
Oil palm sap/wine: The oil palm sap/wine is the exudates that flow when the palm is tapped (Tulley, 1964). Just like the sap of other palms like Raphia, researches conducted by authors like Bassir (1962), Bassir (1968), Obahiagbon and Oviasogie (2007) and Obahiagbon et al. (2007) have shown that the sap contains numerous phytonutrients which plays significant roles in human health. In short, the sap has medicinal role in the cure of malaria, measles, jaundice and the flow of breast milk in nursing mothers (Bassir, 1968). In Nigeria, the drinking of palm wine signals the start and end of all social activities particularly in the rural areas. The palm wine/sap is drunk by over 50 million people in Africa (Obahiagbon and Osagie, 2007).
CONCLUSION
Elaeis guineensis jacq. is the African oil palm which originated from West Africa. The propagation is either by seed or by tissue culture. The palm produces two types of oils: the palm oil from its mesocarp and the kernel oil from its kernel. The palm oil is made-up of both minor and major components. The triglycerides forms the major components while the mono- and diglycerides are in small proportions. The minor components include: the carotinoids, Vitamin E (tocopherols and tocotrienols), phospholipids, squalene, phenolic acids, flavonoids, co-enzyme Q10, chlorophyll, phosphatides, alcohols and sterols. The crude palm oil could be modified to produce products which are characteristically different from the original crude palm oil. Animals and human trials have revealed the significant roles of palm oil components in health. The β carotenes protects against blindness and carcinogens. The tocotrienols are capable of inhibiting cholesterol synthesis (thus lowering the serum cholesterol) and produces inhibitory action on human breast cancer cells. The tocophenols and tocotrienol act as antioxidants thus protecting the body tissues against oxidative damage. Adminstration of Tocotrienol Rich Fraction (TRF) in animals is capable of producing cadio-protective ability. Every part of the oil palm has economic and domestic values. The oils are useful for domestic, industrial and in pharmaceutical preparations. The exudates (palm wine) from the palm have social, economic and health significances.
ACKNOWLEDGMENT
I wish to thank the authors sited and the Management of the Nigerian Institute for oil Palm Research, for their Library used for this review.
REFERENCES
- Ahmad, A.L., C.Y. Chan, S.R. Abd Shukor and M.D. Mashitah, 2010. Adsorption chromatography of carotenes from extracted oil of palm oil mill effluent. J. Applied Sci., 10: 2623-2627.
CrossRefDirect Link - Bassir, O., 1962. Observations on the fermentation of palm wine. West Afr. J. Biol. Applied Chem., 6: 20-25.
Direct Link - Budin, S.B., N.F. Rajab, K. Osman, A.G.M. Top, W.N.W. Mohammed, M.A. Bakar and J. Mohamed, 2006. Effects of palm vitamin E against oxidative damage in Streptozotocin-induced diabetic rats. Malaysian J. Biochem. Mol. Biol., 13: 11-17.
Direct Link - Rukmini, C., 1994. Red palm oil to combat vitamin A deficiency in developing countries. Food Nutr. Bul., 15: 126-129.
Direct Link - Edem, D.O., 2002. Palm oil: Biochemical, physiological, nutritional, hematological and toxicological aspects: A review. Plant Foods Hum. Nutr., 57: 319-341.
CrossRefDirect Link - Das, S., I. Lekli, M. Das, G. Szabo and J. Varadi et al., 2008. Cardioprotection with palm oil tocotrienols: Comparision of different isomers. Am. J. Physiol. Heart Cirl. Physiol., 294: 70-78.
CrossRefPubMedDirect Link - Obahiagbon, F.I. and P. Oviasogie, 2007. Changes in the physico-chemical characteristics of processed and stored Raphia hookeri palm sap (shelf life studies). Am. J. Food Technol., 2: 323-326.
CrossRefDirect Link - Abulude, F.O., M.O. Ogunkoya1, R.F. Ogunleye, A.O. Akinola and A.O. Adeyemi, 2007. Effect of palm oil in protecting stored grains from Sitophilus zeamais and Callosobruchus maculatus. J. Entomol., 4: 393-396.
CrossRefDirect Link - Abong, G.O., M.W. Okoth, J.K. Imungi and J.N. Kabira, 2011. Losses of ascorbic acid during storage of fresh tubers, frying, packaging and storage of potato crisps from four Kenyan potato cultivars. Am. J. Food Technol., 6: 772-780.
CrossRefDirect Link - Inocent, G., D.M. Adelaide, E.L. Gisele, M.O.R. Solange, E.A. Richard and F. Elie, 2011. Impact of three cooking methods (steaming, roasting on charcoal and frying) on the beta-carotene and vitamin c contents of plantain and sweet potato. Am. J. Food Technol., 6: 994-1001.
CrossRef - Hayes, K.C. and P. Khosla, 1992. Dietary fatty acid thresholds and cholesterolemia. EASED J., 6: 2600-2607.
CrossRefPubMedDirect Link - Hendrich, S., K.W. Lee, X. Xu, H.J. Wang and P.A. Murphy, 1994. Defining food components as new nutrients. J. Nutr., 124: 1789S-1792S.
CrossRefPubMedDirect Link - Law-Ogbomo, K.E. and R.K.A. Egharevba, 2006. The use of vegetable oils in the control of Callosobruchus maculatus (F) (Coleoptera: Bruchidae) in three cowpea varieties. Asian J. Plant Sci., 5: 547-552.
CrossRefDirect Link - Krinsky, N.I., 1992. Mechanism of action of biological antioxidants. Proc. Soc. Exp. Biol. Med., 200: 248-254.
CrossRefPubMedDirect Link - Levin, M.S. and A.E. Davis, 1997. Retinoic acid increase cellular retinol binding protein II MRNA and retinol uptake in the human intestinal Caco-2-cell line. J. Nut., 127: 13-17.
CrossRefDirect Link - Yusoff, M.Z.M., M.A. Hassan, S. Abd-Aziz and N.A.A. Rahman, 2009. Start-up of biohydrogen production from palm oil mill effluent under non-sterile condition in 50 L continuous stirred tank reactor. Int. J. Agric. Res., 4: 163-168.
CrossRefDirect Link - McIntyre, B.S., K.P. Briski, A. Gapor and P.W. Sylvester, 2000. Antiproliferative and apoptotic effects of tecopherols and tocotrienols on preneoplastic and neoplastic mouse mammary epithelial cells. Proc. Soc. Exp. Biol. Med., 224: 292-301.
CrossRefPubMedDirect Link - Nesaretnam, K., T.H. Koon, K.R. Selvaduray, R.S. Bruno and E. Ho, 2008. Modulation of cell growth and apoptosis response in human prostate cancer cells supplemented with tocotrienols. Eur. J. Lipid Sci. Technol., 110: 23-31.
CrossRef - Nesaretnam, K., R. Ambra, K.R. Selvaduray, A. Radhakrishnan, K. Reimann, G. Razak and F. Virgili, 2004. Tocotrienol-rich fraction from palm oil affects gene expression in tumors resulting from MCF-7 cell inoculation in athymic mice. Lipids, 39: 459-468.
CrossRefPubMedDirect Link - Obahiagbon, F. I. and A.U. Osagie, 2007. Sugar and macrominerals composition of sap produced by Raphia hookeri palms. Afr. J. Biotechnol., 6: 744-750.
Direct Link - Ogunkunle, A.O., U. Amoti, A.E. Aghimien and A.E. Isenmila, 1999. Characteristics, classification and agricultural potential of some Niger delta soils in Nigeria. Commun. Soil Sci. Plant Anal., 30: 663-675.
CrossRefDirect Link - Henry, O.H., 2011. Monitoring the free fatty acid level of crude palm oil stored under light of different wavelenghts. Am. J. Food Technol., 6: 701-704.
CrossRefDirect Link - Qureshi, A.A., N. Qureshi, J.J. Wright, Z. Shen and G. Kramer et al., 1991. Lowering of serum cholesterol in hypercholesterolemic humans by tocotrienols (palmvitee). Am. J. Clin. Nutr., 53: 1021S-1026S.
CrossRefDirect Link - Cottrell, R.C., 1991. Introduction: Nutritional aspects of palm oil. Am. J. Clin. Nutr., 53: 989S-1009S.
CrossRefDirect Link - Roo, B.S.N., 2000. Potential use of red palm oil in combating vitamin A deficiency in India. Food Nutr. Bull., 21: 202-211.
CrossRefDirect Link - Goh, S.H., Y.M. Choo and S.H. Ong, 1985. Minor constituents of palm oil. J. Am. Oil Chem. Soc., 62: 237-240.
CrossRefDirect Link - Goh, S.H., H.T. Khor and P.T. Gee, 1983. Phospholipids of palm oil (Elaeis guineensis). J. Am. Oil Chem. Soc., 59: 296-299.
CrossRef - Sambanthamurthi, R., K. Sundram and Y.A. Tan, 2006. From biowaste to bioproducts: Phenolics antioxidants from oil palm waste. Proceedings of the 23rd International Conference on Polyphenols, August 22-25, 2006, University of Manitoba, Winnipeg, Canada.
Direct Link - Song, B.L. and R.A. DeBose-Boyd, 2006. Insig-dependent ubiquitination and degradation of 3-hydroxy-3-methylglutaryl coenzyme a reductase stimulated by δ- and γ-tocotrienols. J. Biol. Chem., 281: 25054-25061.
CrossRefPubMedDirect Link - Starks, M.A., S.L. Starks, M. Kingsley, M. Purpura and R. Jager, 2008. The effects of phosphatidylserine on endocrine response to moderate intensity exercise. J. Int. Soc. Sports Nutr., Vol. 5.
CrossRefDirect Link - Chong, Y.H. and T.K. Ng, 1991. Effects of palm oil on cardiovascular risk. Med. J. Malaysia, 46: 41-50.
PubMedDirect Link - Zeisel, S.H., D. Char and N.F. Sheard, 1986. Choline, phosphatidylcholine and sphingomyelin in human and bovine milk and infant formulas. J. Nutr., 116: 50-58.
CrossRefPubMedDirect Link








