Short Communication
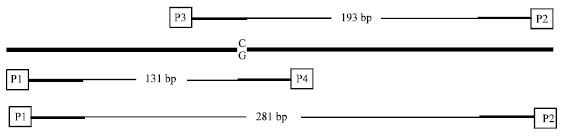
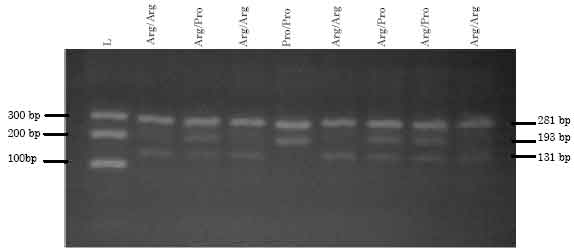
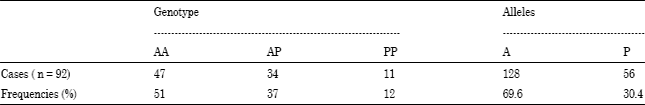
Detection of Arg72Pro Polymorphism of the Tumor Suppressor Gene (TP53) by a Rapid One-step Tetra-primer Amplification Refractory Mutation System-PCR
Department of Analytical Chemistry, Faculty of Pharmacy, University of Aleppo, Aleppo, Syria
Amal Alachkar
Department of Pharmacology, Faculty of Pharmacy, University of Aleppo, Aleppo, Syria