Research Article
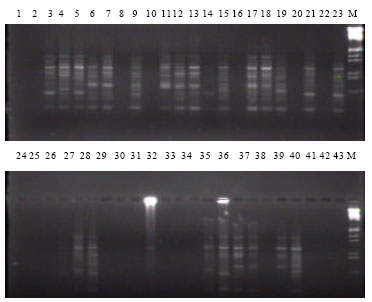
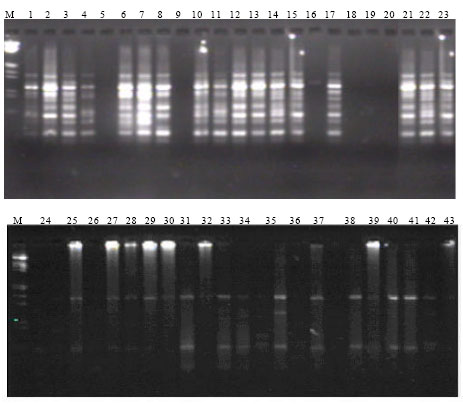
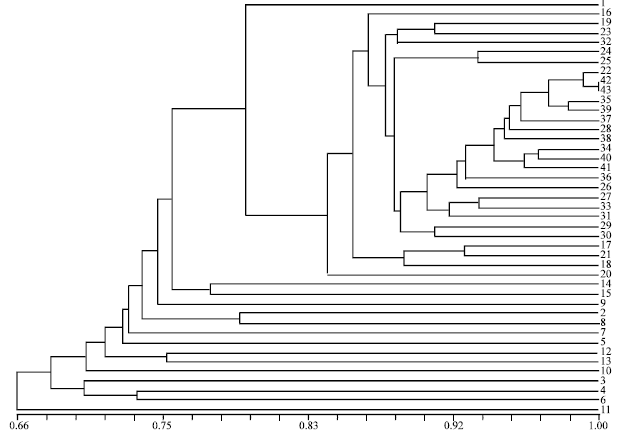
Assessment of Genetic Diversity and its Usefulness for Varietal Identification in Indian Elite Varieties of Wheat (T. aestivum L. em thell.) Using RAPD Markers
Molecular Biology Unit, Institute of Medical Sciences, Banaras Hindu University, India
Sanjay K. Jaiswal
Department of Genetics and Plant Breeding, Institute of Agricultural Sciences, Banaras Hindu University, Varanasi, India
Manish K. Vishwakarma
Department of Genetics and Plant Breeding, Institute of Agricultural Sciences, Banaras Hindu University, Varanasi, India
LiveDNA: 91.17474
Ved P. Rai
Department of Genetics and Plant Breeding, Institute of Agricultural Sciences, Banaras Hindu University, Varanasi, India
Priti Upadhyay
Department of Genetics and Plant Breeding, Institute of Agricultural Sciences, Banaras Hindu University, Varanasi, India
Arun K. Joshi
Department of Genetics and Plant Breeding, Institute of Agricultural Sciences, Banaras Hindu University, Varanasi, India