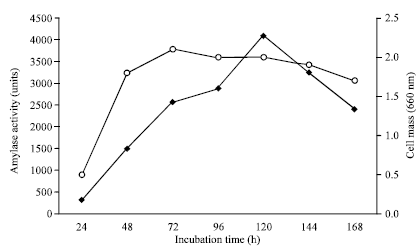
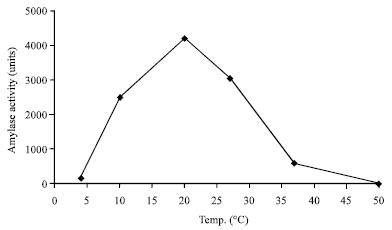
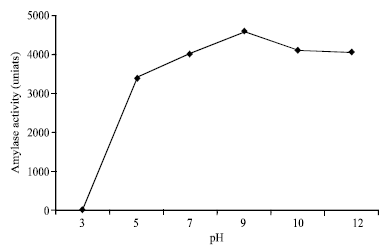
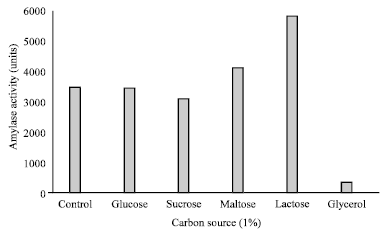
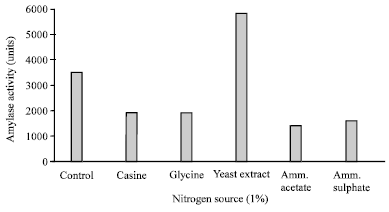
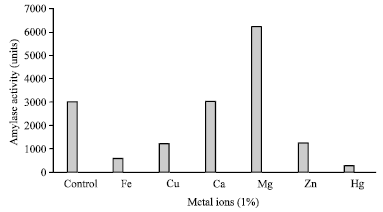
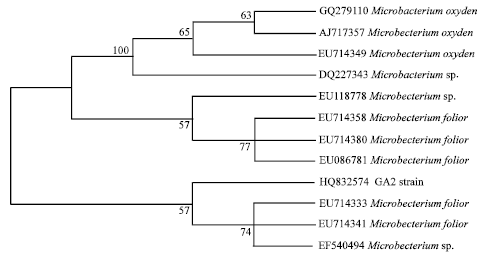
Research Article
Production of Cold-active Extracellular α-Amylase by Newly Isolated Microbacterium foliorum GA2 from Gangotri glacier, Western Himalaya, India
Protein Research Laboratory, Department of Biotechnology and Microbiology, Integral University, Lucknow-226026, India
M. Kuddus
Protein Research Laboratory, Department of Biotechnology and Microbiology, Integral University, Lucknow-226026, India
I.Z. Ahmad
Protein Research Laboratory, Department of Biotechnology and Microbiology, Integral University, Lucknow-226026, India
J.M. Arif
Protein Research Laboratory, Department of Biotechnology and Microbiology, Integral University, Lucknow-226026, India