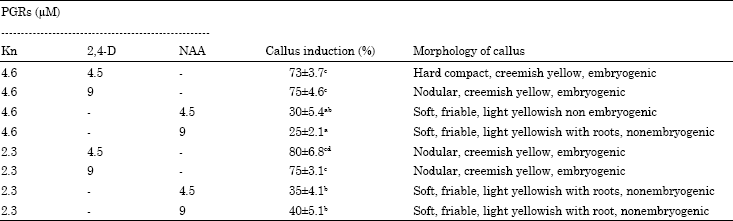
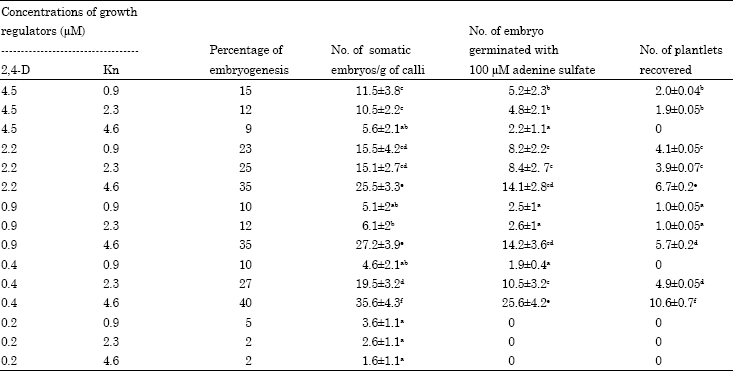
Research Article
Somatic Embryogenesis in Swertia chirata Buch. Ham. ex Wall. - A Multipotent Medicinal Herb
Laboratory of Plant Biotechnology, Department of Botany, Presidency College, Kolkata-700073, India
LiveDNA: 91.19935
Arpita Dafadar
Laboratory of Plant Biotechnology, Department of Botany, Presidency College, Kolkata-700073, India
Rituparna Kundu Chaudhuri
Bethune College, Kolkata-700006, India