Research Article
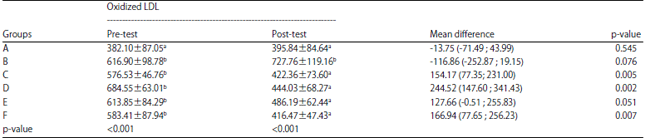
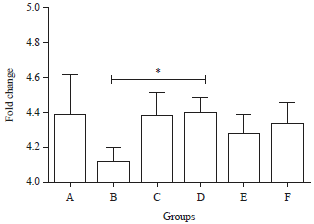
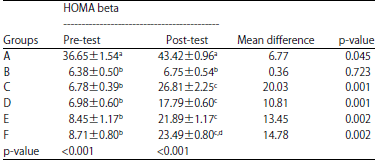
Effects of 7-Hydroxy-2-(4-hydroxy-3-methoxyphenyl)-chromen-4 -one from Swietenia macrophylla King Seed on Oxidized LDL, HOMA Beta and Glucagon like Peptide 1 (GLP-1) Gene Expression in Type 2 Diabetic Rats
Department of Biochemistry, Faculty of Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia
LiveDNA: 62.16993
Sunarti
Department of Biochemistry, Faculty of Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia
Ahmad Hamim Sadewa
Department of Biochemistry, Faculty of Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia
Sri Mursiti
Department of Mathematics and Sciences, Universitas Negeri Semarang, Semarang, Indonesia
Mustofa
Department of Pharmacology, Faculty of Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia