Research Article
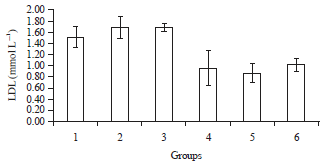
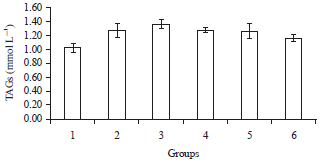
Phytochemical, Antihyperglycaemic and Lipid Profile Effects of Methanol Extract Fraction of Ricinus comminus Seeds in Alloxan Induced Diabetic Male Wistar Albino
Department of Biochemistry, Faculty of Biological Sciences, University of Nigeria, Nsukka, Enugu State, Nigeria
C. Nweje-Anyalowu Paul
Department of Biochemistry, Faculty of Biological Sciences, University of Nigeria, Nsukka, Enugu State, Nigeria
E. Joshua Parker
Department of Biochemistry, Faculty of Biological Sciences, University of Nigeria, Nsukka, Enugu State, Nigeria
LiveDNA: 234.4382
Uroko Robert Ikechukwu
Department of Biochemistry, Faculty of Biological Sciences, University of Nigeria, Nsukka, Enugu State, Nigeria
LiveDNA: 234.16532