Research Article
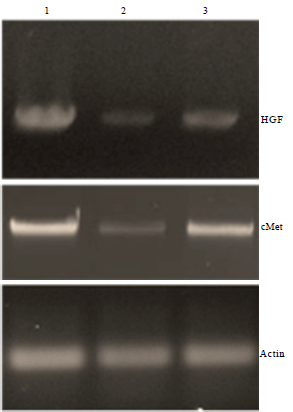
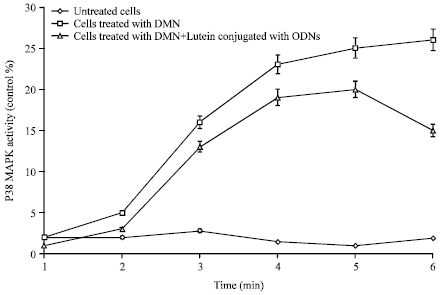
Effect of Bioconjugation of Oligodeoxynucleotides with Lutein on N-nitrosodimethylamine Induced Fibrosis in Cultured Human Retinal Pigment Epithelial Cells
Department of Biochemistry, JSS College of Arts, Commerce and Science (Autonomous), Ooty Road, Mysore-570025, Karnataka, India
Kondagula Shivappa Swetha
Department of Biochemistry, JSS College of Arts, Commerce and Science (Autonomous), Ooty Road, Mysore-570025, Karnataka, India