Short Communication
Oxidative Stress In Anti Thyroperoxidase Antibody Positive Hypothyroid Patients
Department of Biochemistry, Pondicherry Institute of Medical Sciences, Puducherry, India
Zachariah Bobby
Department of Biochemistry, Jawaharlal Institute of Postgraduate Medical Education and Research, Puducherry, India
Abdoul Hamide
Department of Biochemistry, Jawaharlal Institute of Postgraduate Medical Education and Research, Puducherry, India









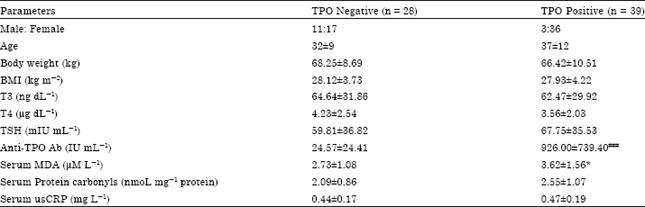
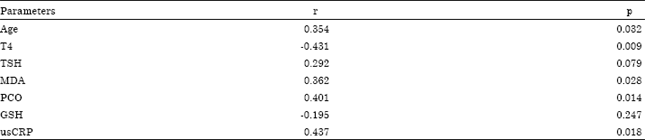
Anwar Alashtal Reply
Hi,
As per the below report my TPOAb is above 600 IU/ML in AUG 2009, I was supposed to take 50MCG on daily basis but i did not till date. No clear phisical signs of the problem in my body till date. What do you recomend
Reagards.
Anwar
Report from Anand Diagnostic Laboratory
Dated 11/8/2009
ANTITHYROPEROXIDASE ANTIBODY
REFERENCE RANGE – UPTO 34 IU/ML
OBSERVED VALUES – MORE THAN 600 IU/ML
Nivedita Nanda
When your TPO level is so high it clearly indicates you have to follow medications as instructed by the physician. Our body has a great tolerance limit. Therefore it takes time before physical signs of these problems are detected. one should always adhere to the medicines and the exact dose instructed by the physician and the medicine dosage should be altered as per clinician's instructions.
bernie mchugh Reply
Iwas on eltroxin 100mcg/50mcg alt days for 10 years.
6 months ago i went hyper. now i am taking neo-mercazole 20.
my anti-thyroperoxidase is now >1000.00
i feel sick after taking neo-mercazole.
my dr.tells me to keep taking it.
felt good before i started to take it.
any idea what i should do.
bernie