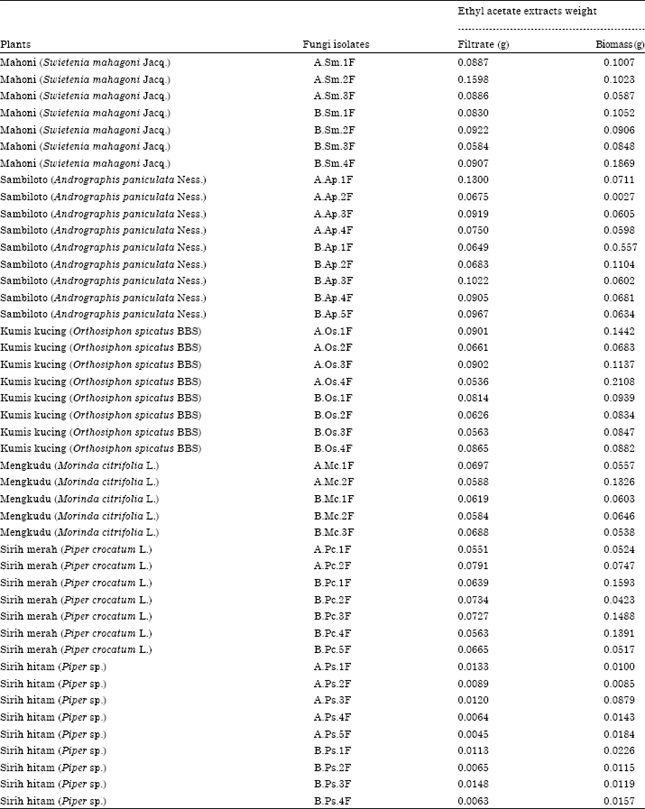
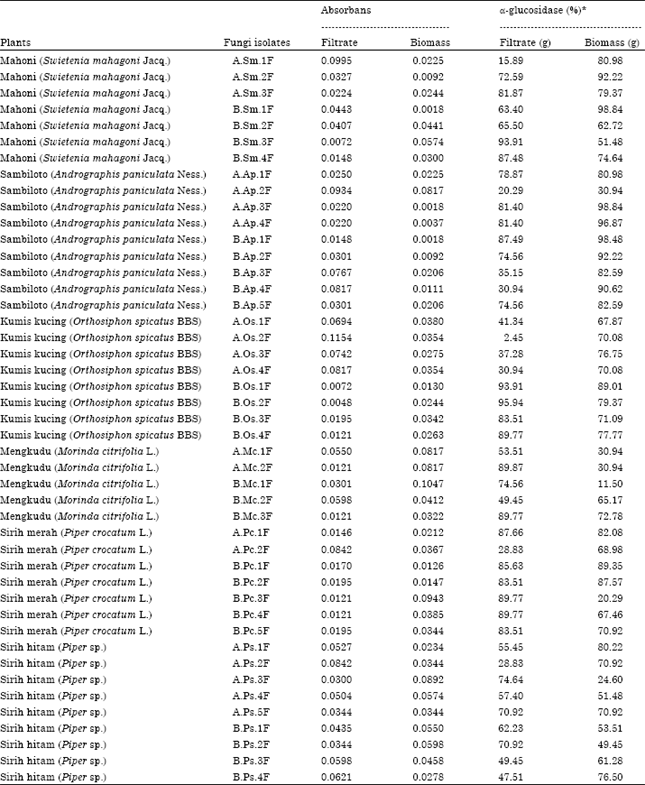
Research Article
Potential Endophytic Microbes Selection for Antidiabetic Bioactive Compounds Production
Departemnt of Chemistry, Faculty of Mathematics and Natural Sciences, University of Indonesia, Depok, West Java, 16911, Indonesia
Yoice Srikandace
Research Center for Biotechnology Indonesian Institute of Sciences (LIPI), Jl. Raya Bogor KM 46 West Java, 16911, Indonesia
Wahyudi Priyono Suharso
Departemnt of Chemistry, Faculty of Mathematics and Natural Sciences, University of Indonesia, Depok, West Java, 16911, Indonesia
Herry Cahyana
Departemnt of Chemistry, Faculty of Mathematics and Natural Sciences, University of Indonesia, Depok, West Java, 16911, Indonesia
Partomuan Simanjuntak
Research Center for Biotechnology Indonesian Institute of Sciences (LIPI), Jl. Raya Bogor KM 46 West Java, 16911, Indonesia