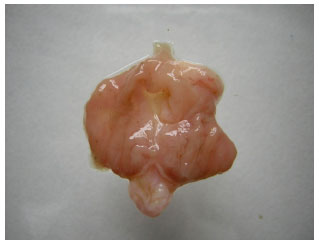
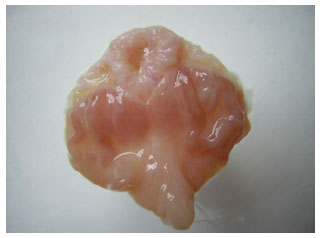
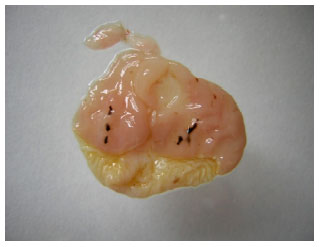
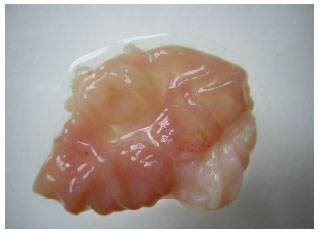
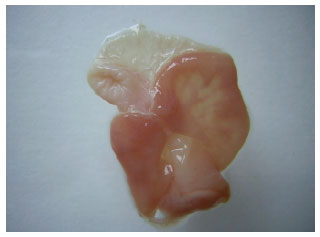
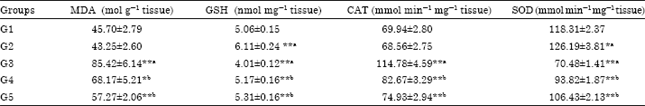
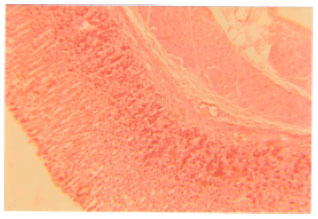
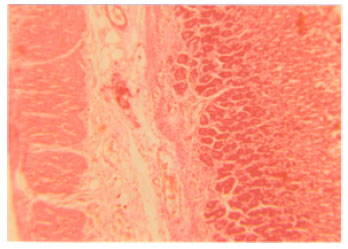
Research Article
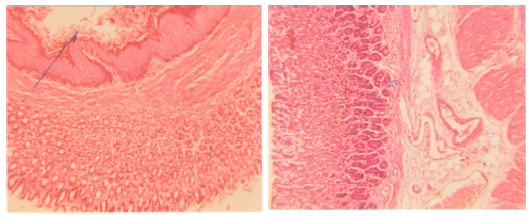
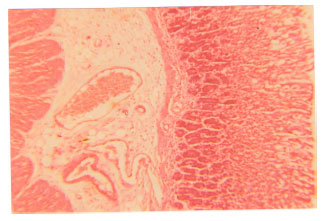
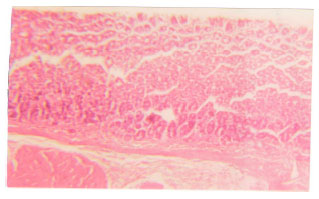
Antiulcer Effects of Alpha Lipoic Acid on Gastric Acid Secretion and Mucosal Defense Factors in Rats
National Research Centre, Department of Biochemistry, 33 El-Tahrir Street, Dokki 12622, Cairo, Egypt
M.M. Ali
National Research Centre, Department of Biochemistry, 33 El-Tahrir Street, Dokki 12622, Cairo, Egypt
N.M. El-Sammad
National Research Centre, Department of Biochemistry, 33 El-Tahrir Street, Dokki 12622, Cairo, Egypt
M.A. El-Shaer
National Research Centre, Department of Pathology, 33 El-Tahrir Street, Dokki 12622, Cairo, Egypt