Research Article
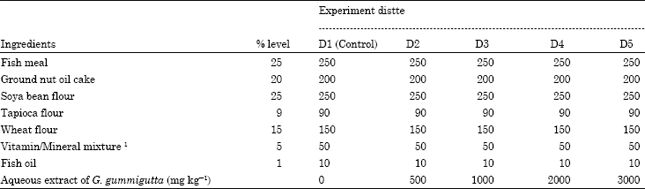
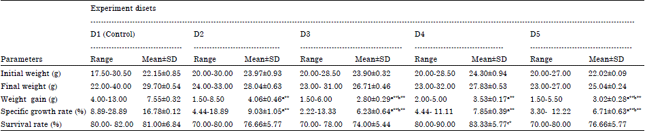
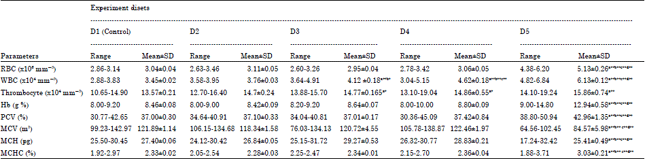
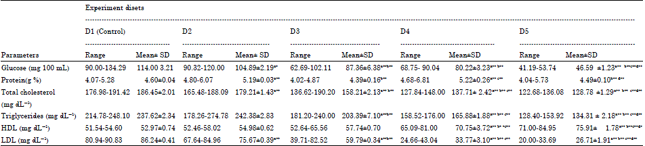
Effect of Fruit Rind Extract of Garcinia gummi-gutta on Haematology and Plasma Biochemistry of Catfish Pangasianodon hypophthalmus
Laboratory of Conservation Biology, Department of Zoology, University of Kerala, Kariavattom, Thiruvananthapuram 695 581, Kerala, India
G.L. Priyanka
Laboratory of Conservation Biology, Department of Zoology, University of Kerala, Kariavattom, Thiruvananthapuram 695 581, Kerala, India