Research Article
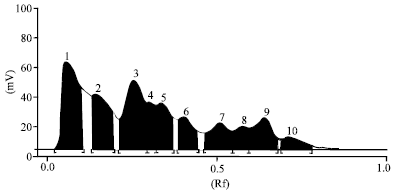
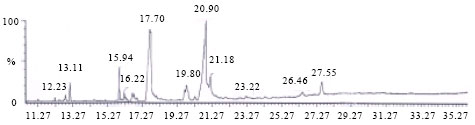
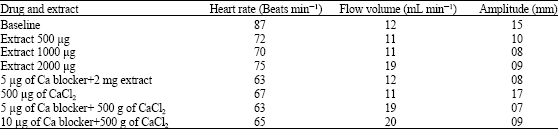
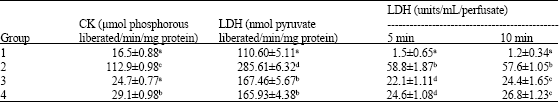
Interpretation of Inotropic Effect Exhibited by Desmodium gangeticum Chloroform Root Extract Through GSMS and Atomic Mass Spectroscopy: Evaluation of its Anti Ischemia Reperfusion Property in Isolated Rat Heart
Department of Medical Biochemistry, School of Chemical and Biotechnology, Sastra University, Thirumalaisamudram, Thanjavur, Tamil Nadu, India
R.S.S. Srivats
Department of Biotechnology, Sastra University, India
R. Gomathi
Department of Biotechnology, Sastra University, India
M.M. Shabi
Division of Pharmacology and Toxicology, Centre for Advanced Research in Indian System of Medicine, Sastra University, India
J. Paddikkala
Department of Plant Biotechnology, Amala Cancer Research Center, Malanagar, Trichur, Kerala, India