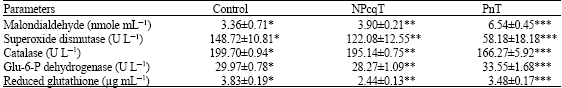
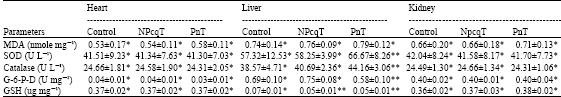
Research Article
Impact of Plasmodium berghei and Chloroquine on Haematological and Antioxidants Indices in Mice
Department of Biochemistry, Ambrose Alli University, Ekpoma, Nigeria
A.O. Onigbinde
Department of Biochemistry, Ambrose Alli University, Ekpoma, Nigeria