Research Article
Selenium and α-Difluromethylornthine in Combination have Strong Activity Against Elevated Polyamines and Glucose Levels in Serum
Department of Chemistry, College of Science, King Faisal University, Saudi Arabia
Polyamines such as spermidine, spermine and their precursor, putrescine are extensively distributed in nature and display a variety of important biological activities (Blagbrough et al., 1997). These compounds influence DNA replication and translation, protein synthesis, membrane stabilization and the activity of certain kinases and topoisomerases. Charge neutralization of intracellular DNA and RNA may be among the most important physiological roles of these compounds. Stabilization of precise DNA conformations may be significant for nucleosome arrangement, chromatin condensation and gene expression (D` Agostino et al., 2005; Tabor and Tabor, 1999; Mattews, 1993).
In mammalian cells, polyamines are synthesized from L-arginine to L-ornithine using arginase in extrahepatic tissues (Méndez, 1995). Ornithine is then decaboxylated into putrescine by the action of ornithine decarboxylase enzyme (ODC, EC 4. 1.1.17) (Ramya et al., 2006; McCann and Pegg, 1992; Heby, 1981). ODC is the primary and rate restrictive enzyme in polyamine biosynthesis. Increased ODC activity are parallel to levels of polyamines generally be increasing the proliferating cells such as those live in embryos and cancer cells (Krause et al., 2000; Pegg et al., 1987). Therefore, inhibition of ODC activity might stimulate the reduction of intracellular polyamines, which an effective anticancer cure plans.
Alpha-Difluromethylornthine (DFMO) is an ornithine analogue that inhibits ODC enzyme activity irreversibly (Manni et al., 2007; Metcalf et al., 1978). In the presence of both ODC and DFMO a transitional carbonic species is generated from the decarboxylation development to DFMO by an enzymatic system. With the loss of fluorine the transitional carbonic species alkylates a nucleophlic residue in nearby at the active site, resulting in covalent binding of inhibitor to the enzyme (Metcalf et al., 1978). Therefore, DFMO has been established to be considerably repressing many cancer formations (Meyskens and Gerner, 1999).
Selenium is an indispensable trace element in the nutrition for humans and other animals and is necessary for the development of mammalian cells in way of life (Zeng, 2002). Sodium selenate has insulin like effects in vitro in fat cells which causes the translocation of glucose transporters to the plasma membrane (Ezaki, 1990). The metabolic basis of this nutritional purpose remained unclear, however, until it was identified that the enzyme glutathione peroxidase posses Se as a vital element in its catalytic center (Rotruck et al., 1973). The proteins which include Se in its structure, such as, glutathione peroxidases and thioredoxin reductases, are important antioxidant and detoxification agents (Ganther, 1999). The relation between polyamines biosynthesis and Se metabolism is that both of them required S- adenosylmethionene as a cofactor (Kajander et al., 1990).
Putrescine and spermidine are necessary for in vitro insulin and protein biosynthesis, whereas spermine depletion affects numerous processes implicated in insulin metabolism. It is usually agreed that glucose is the most important control device of the insulin gene (Welsh, 1989). This nutrient is thought to enhance insulin mRNA contents both in vitro and in vivo and increases of as much as 10-fold have been observed on glucose stimulation (Brunstedt and Chan, 1982). The glucose consequence is mediated by a combination of enlarged transcription of the insulin gene and a careful stabilization of insulin mRNA against degradation (Welsh et al., 1985). The stimulatory effect of glucose on insulin-gene transcription may, in part, be mediated by cyclic AMP (Nielsen et al., 1985). It has been shown that other nutrients also improve insulin-gene expression (Welsh et al., 1986) and that the insulin mRNA contents often correlate well with the rate of islet ATP creation (Spinas et al., 1987; Eizirik et al., 1988).
The aim of present research was to study the effects of Se and/ or DFMO on the high levels of polyamine and glucose levels of induced in the serum of experimental mice utilizing both L-arginine and L-ornithine.
MATERIALS AND METHODS
Chemicals
All the chemicals used were AR grade and obtained from Sigma chemical Co and BDH chemicals LTD.
Methods
DFMO dosing solution was dissolved in deionized water and delivered in 0.2 mL. Sodium selenite (Na2SeO3) was prepared with the required amount in deionized water and stored at 4 °C. DFMO and sodium selenite were administered individually once daily by intraperitonealy (ip) injection.
Animals
Female Swiss albino mice (kindly provided by Veterinary Research Centre and Animal Production, King Faisal University, Saudi Arabia) at 6 weeks of age, 24 ± 2 g body weight (b.wt.). They were permitted free admission to feed a standard diet and water with a 12 h light/ dark cycle at controlled temperature.
Study Groups and Sampling
Polyamines levels in the experimental mice were elevated by adding together both L- arginine (2%) and L-ornithine (2%) in the drinking water for 4 weeks according to the method described by Teixeira et al. (2002). The mice were randomly distributed into seven groups (each group n = 8), housed separately in stainless steel cages.
| Group 1 | : | Received water free of any chemical additives |
| Group 2 | : | Received L-arginine (2%) and L-ornithine (2%) in drinking water |
| Group 3 | : | As group 2 and received seven doses of DFMO (2 mg kg-1 b.wt.) by ip injection |
| Group 4 | : | As group 2 and received seven doses of Se (0.5 mg kg-1 b.wt.) by ip injection |
| Group 5 | : | As group 2 and received seven doses of both Se (0.5 mg kg-1 b.wt.) and DFMO (2 mg kg-1 b.wt.) by ip injection |
| Group 6 | : | As group 2 and received seven doses of Se (1 mg kg-1 b.wt.) by ip injection |
| Group 7 | : | As group 2 and received seven doses of both Se (1 mg kg-1 b.wt.) and DFMO (2 mg kg-1 b.wt.) by ip injection |
Homogenate Preparation
The experimental animals were anesthetized and blood samples were collected. The blood samples were centrifuged at 3000 rpm for 10 min. Serum samples were collected for biochemical parameter determinations.
Estimation of Glucose
Glucose concentration in the serum was measured. In this method glucose is oxidized by glucose-oxidase to gluconic acid and hydrogen peroxide, followed by the reaction of hydrogen peroxide with 4-aminophenazone and phenol, catalyzed by phenol oxidase to produce a complex dye that is registered at 540nm.
Estimation of Cholesterol
Cholesterol concentration was measured in the serum by cholesterol oxidase. The total cholesterol concentration is proportional to the dye product formed by the reaction of hydrogen peroxide released with the 4-aminophenazone and phenol reagent, measured at 540nm.
Estimation of Triglycerides
Triglyceride levels were determined in the serum. This method is based in the triglyceride hydrolysis by lipase and the glycerol formed is utilized by glycerol kinase, phosphoglycerol oxidase and peroxidase to form hydrogen peroxide, which reacts with 4-aminophenazone and phenol to produce a complex that is measured at 620nm.
Polyamine Determinations
Aliquots of serum were extracted with 0.6 N perchloric acid for 1 h at 4 °C prior to centrifuge at 10000 rpm for 15 min. The supernatant was used for polyamine determination. The polyamine levels were determined using high-pressure liquid chromatography as described by Manni et al. (2002). The concentration of polyamines was determined by graphical analysis relative to that obtained from a standard curve generated for each polyamine.
Statistical Analysis
All the values are represented as Mean ± SD (n = 8). Student’s t-test was applied to calculate the significance of difference between groups. The level of significance was set at p < 0.05.
Effect of L- Arginine and L- Ornithine Supplementation
Arginine is a substrate for ornithine biosynthesis and the ornithine is the originator of the polyamines. Therefore, the consequence of addition of 2% L-arginine and 2% L-ornithine on the polyamines synthesis were demonstrated. Body weights of mice treated with L-arginine and L-ornithine were not significantly affected in all studied groups in the experiment. The levels of putrescine, spermidine and spermine in the serum of group 1 (not treated with L-arginine or L-ornithine) were increased significantly (p < 0.001) from 3.67 ± 0.53, 3.22 ± 0.61 and 2.63 ± 0.23 to 6.05 ± 0.42, 4.39 ± 0. 44 and 3.34 ± 0.11, respectively compared to the serum values in the same order (Table 1). These results indicate that the addition of both L-arginine and L-ornithine to the drinking water of the experimental animals can cause a significant elevation in the polyamine levels in serum.
Effect of Se or/and DFMO on the Elevated Polyamine Levels
The supplementation of experimental animals with 0.5 mg Se kg-1 body weight (Table 1, group 4) reduce the elevated level of putrescine in serum by 14.2%. Also, high Se dose 1 mg kg-1 body weight (Table 1, group 6) decrease the putrescine level in serum by 32.2%. Group 2 was considered as a control. As represented in Table 1, group 4 low Se dose reduced the spermidine level in serum by 12.8%. Moreover, the high Se dose (Table 1, group 6) depleted the spermidine level in serum by 22.3%. The spermine level in serum was decreased by 3.6% with low Se dose and 15.6% with high Se dose.
As represented in Table 1, group 3 the DFMO abolished both the putrescine levels in serum by 77.4% and the spermidine level by 62.2 %. On the other hand, the DFMO increase the spermine level 22.5% in serum. The combination between both Se and DFMO tend to normalize the polyamine levels in the serum.
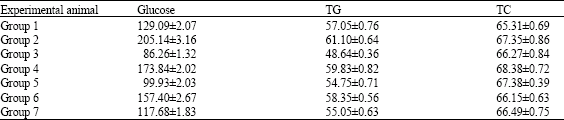
Glucose, Triglycerides and Cholesterol Levels in Serum
The supplementations with 2% L-arginine and 2% L-ornithine increased the glucose levels in the serum of group 2 significantly (p < 0.05) compared to untreated control group 1. Se or DFMO caused depletion in the elevated glucose concentrations as represented in Table 2. Se and DFMO administrations interperitoneally tended to normalize the elevated serum glucose levels (Table 2).
The triglyceride levels in the serum did not change greatly and no differences were observed between the various experimental groups except group 3 that treated with DFMO which declined the triglyceride level by 20.4% than the control group (Table 2). Also, the levels of cholesterol in serum revealed no significant differences between the various experimental mice groups (Table 2).
| Table 1: | Level of polyamines such as putrescine, spermidine and spermine in serum of the control and experimental mice |
 | |
| Table 2: | Levels of glucose, TG and TC in serum of the experimental animals |
 | |
DISCUSSION
In the present study polyamines and glucose concentrations increased significantly with supplementation of both L-arginine and L-ornithine in the drinking water of the experimental mice. L-Arginine and L-ornithine, as a target for consumption of arginase activity, are essential for the synthesis of polyamines (Wu and Morris, 1998). Arginase activity is linked to cell development and connective tissue formation, which is connected with polyamines, proline and in ammonia detoxification. Many reports stated that arginine starvation greatly decreases putrescine, spermidine and spermine contents in the rate (Schertel and Eichler, 1991) mice (Teixeira et al., 2002).
Polyamines levels inside the cells are firmly synchronized and can be altered through the putrescine, spermidine and spermine biosynthesis and interconversation, particularly ornithine decarboxylase (Ackermann et al., 2003), that quickly responds to a number of stimuli and are largely linked with cell growth (Wallace et al., 2003; Thomas and Thomas, 2001).
In ordinary tissues ODC action is enlarged by a multiplicity of ecological and hereditary factors linked with carcinogenesis, including ultraviolet light and carcinogenic agents. Increased ODC activity persists and is associated with a wide variety of epithelial neoplasms including skin, breast, prostate, colon, (Gerner and Meyskens, 2004). Polyamines and ODC are increased in breast cancer corresponding to regular breast tissue. The enlarge of polyamines levels are correlated with a less differentiated and more metastatic tumor phenotype (Glikman et al., 1987; Canizares et al., 1999). Increased polyamine synthesis has been associated with proliferation and progression of breast cancer and thus, is a potential target for anticancer therapy (Satriano et al., 1998). Polyamine depletion by DFMO has been shown to decrease pulmonary and bone metastasis from human breast cancer cell (McCann and Pegg, 1992; Davidson et al., 1999).
The toxic dose of DFMO is 200 and 135 mg kg-1 in rats and rabbits, respectively (Kirchner et al., 1999). In the present study we utilized 2 mg DMFO kg-1 body weight of the experimental mice. DFMO inhibits putrescine and spermidine and increases the level of spermine, these results are in agreement with the results of Jun et al.(2007) and Halline et al. (1989).
The results in the present study indicates that the elevated polyamines levels can be modulated with combination of both Se and DFMO.
Battell et al. (1998) applied very high selenium doses over 2 mg kg-1 i.e., close to the LD50, which are equal to 3.5 mg kg-1 body weight per day to achieve decreased blood glucose level. In the present study we applied low dose of Se (0.5 mg kg-1) and high dose of Se (1 mg kg-1) body weight. Selenium supplementation has in recent times been shown to decrease total cancer incidence. The results indicts that the elevated polyamine levels can be modulated with combination of both Se and DFMO. However, the mechanism of action of selenium as an anticarcinogenic agent has yet to be elucidated (Redman et al., 1998). Earlier studies in animals and humans have shown that selenium compounds can prevent cancer development. The results suggest that selenium is able to reduce the risk for liver cancer even when it is used only during a short period of time covering the promotion phase of the carcinogenic process. Chemically induced hepatocarcinogenesis may be prevented by selenium supplementation both during promotion and progression phase (Bergman et al., 2005).
Present results are in agreement with the results of Battell et al. (1998) in that Se compounds induce decrease in the blood glucose level. Elevated blood glucose levels was reduced 50- 80% following Se administration to type I diabetic rats. Se action in high polyamine levels seems to result from reversal of abnormal hepatic expression of glycogenic, gluconeogenic and lipogenic enzymes, particularly glucokinase, pyruvate kinase and glucose 6- phosphate dehydrogenase (Stapleton, 2000). Polyamine cations exist at millimolar concentration in panceriatic β cells.
In the present study the glucose concentration increased in the elevated polyamine levels and we utilized DFMO and Se to attenuate the glucose induced in the elevated polyamines levels. DFMO specifically and irreversibly inhibits the ODC enzyme that present in the β-cells which lead to depletion of putrescine and spermidine (Eiziriket et al., 1988). Very little is known about the effect of both Se and DFMO on the elevated levels of amines poly. In the present study, we have examined whether Se or DFMO or both play a role in polyamine and glucose regulations. In agreement with present results Ramya et al. (2006) reported that the elevated polyamine levels by L arginine and L ornithine did not change the triglycerides or cholesterol levels in serum but increase the glucose concentration in blood.
L-arginine and L-ornithine inhibit glucose oxidation and had no effect on the lipolysis in serum. Although no direct measurements of glucose transport have been conducted the results of a series of experiments indicate that Se and DFMO similar to insulin which facilitate glucose transport in cells. The extent to which insulin and polyamines share a common pathway of action was further studied by evaluating the interaction between spermidine and the insulin receptor site. The relationship between the structures of the polyamines and their insulin-like properties does not, at present, provide any clear insight concerning the mechanism of action of these compounds. Similar structural requirements may occur for the stabilizing effects of polyamines on glucose transport.
The glucose-induced increase the synthesis of insulin mRNA and total RNA remained unchanged in islets treated with DFMO, suggesting that the glucose effect was not mediated by increased spermidine. However, the finding that insulin mRNA levels did not increase, when the glucose-induced rise in both polyamines was prevented, implicates spermine alone, or the combination of spermine and spermidine, as a possible mediator of parts of the glucose effect. It is unlikely that Se in addition to DFMO exerted a general and non-specific effect on the islet RNA (Eizirik et al., 1988) metabolism, since the rates of total RNA synthesis and turnover were not affected by the inhibitors.
The present experiments demonstrate that L-arginine and L ornithine increase blood glucose level. The protective action of polyamines on β-cells was demonstrated, as well as the capability of these compounds to accelerate their replication when they were damaged by alloxan administration. L-arginine and spermidine inhibited hemoglobin glycation and lipid peroxidation in diabetic animal models. The precise mechanism by which L-arginine and spermidine act is still obscure.
In conclusion, present study showed that inhibition of polyamins biosynthesis with DFMO markedly suppressed tumor cells.
ACKNOWLEDGMENT
The financial support by the Deanship of Scientific Research (Project No. 6021) King Faisal University, Saudi Arabia is gratefully acknowledged.