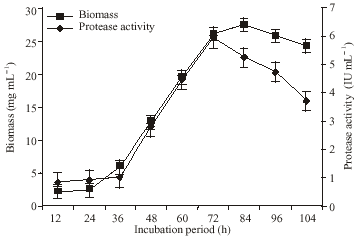
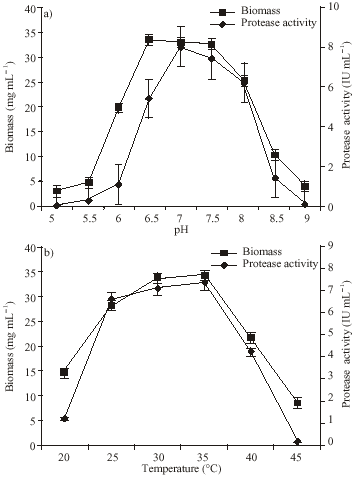
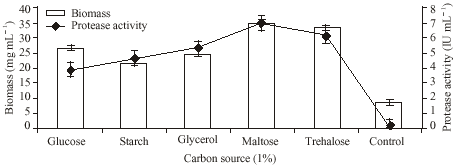
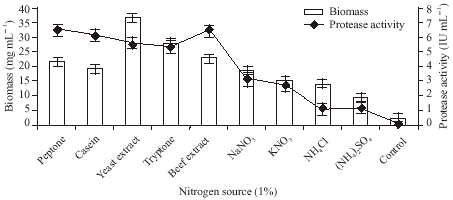
Research Article
Production of Extracellular Protease by Streptomyces albidoflavus
Department of Microbiology, Acharya Nagarjuna University, Guntur-522 510, Andhra Pardesh, India
M. Vijayalakshmi
Department of Microbiology, Acharya Nagarjuna University, Guntur-522 510, Andhra Pardesh, India