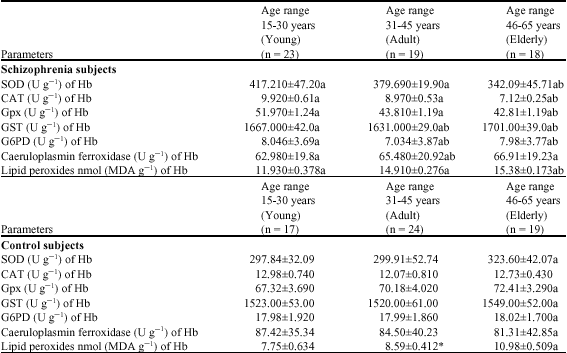
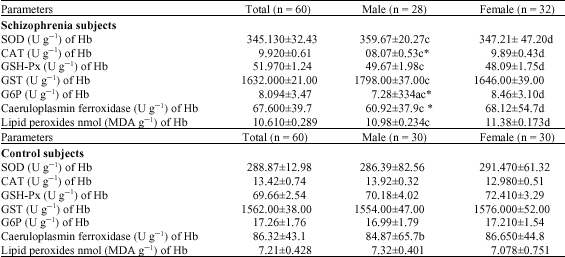
Research Article
Age and Gender Related Changes in Free Radical Pathology and Antioxidant Defense in Schizophrenia
Post Graduate and Research Department of Biochemistry, Dr. N.G.P. Arts and Science College, Coimbatore, Tamilnadu, India
P. Chinnaswamy
Institute of Laboratory Medicines, Kovai Medical Center and Hospitals, Coimbatore, Tamilnadu, India