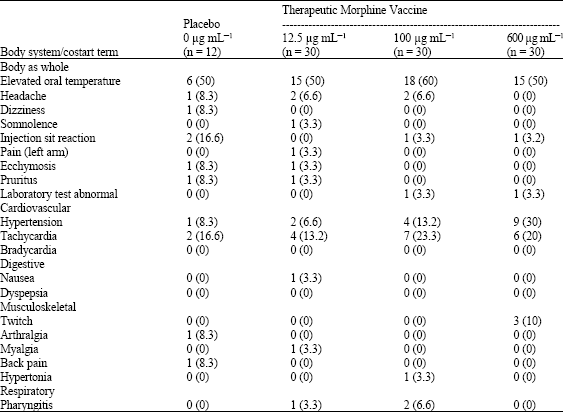
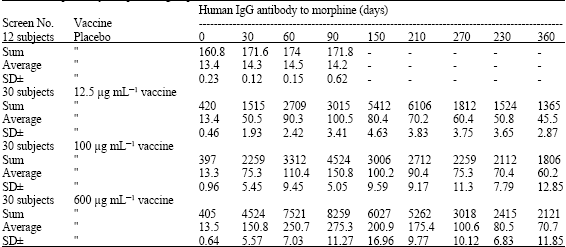
Research Article
Study of Human Therapeutic Morphine Vaccine: Safety and Immunogenicity
Department of Pilot Biotechnology, Pasteur Institute of Iran, Tehran-Iran
D. Norouzian
Department of Pilot Biotechnology, Pasteur Institute of Iran, Tehran-Iran
A. Farhangi
Department of Pilot Biotechnology, Pasteur Institute of Iran, Tehran-Iran
M.R. Mehrabi
Department of Pilot Biotechnology, Pasteur Institute of Iran, Tehran-Iran
M. Bakhtiari
Judicial Ministry, Tehran-Iran
P. Afshar
Judicial Ministry, Tehran-Iran
A. Allahverdi
Department of Pilot Biotechnology, Pasteur Institute of Iran, Tehran-Iran
M.A. Mofidian
S.P.I Institute, Karaj-Iran
A. Shayan
Department of Pilot Biotechnology, Pasteur Institute of Iran, Tehran-Iran