Research Article
Changes in Rabbit Liver Function Markers after Chronic Exposure to Ethanol
Department of Medical Biochemistry, Delta State University, Abraka, Nigeria
E.O. Anosike
Department of Biochemistry, University of Port Harcourt, Nigeria
Alcoholism, one of the most common chronic disorders in our society, causes (or promotes) a plethora of diseases and injuries. Since alcohol is almost entirely detoxified in the liver (Lieber, 1998), hepatic disorders are very common in alcoholics. These disorders range from very mild reversible changes to life-threatening irreversible diseases. However, it has been observed that the manifestation of these diseases is influenced by individual susceptibility, dose and duration of alcohol intake (Wetterling et al., 1999). It has been estimated that the minimum alcohol intake associated with appreciable liver damage is 16 units (about 175 g) of alcohol daily for 5 years (Sherlock, 1995). Diehl (1989) reported that about 30% of alcoholics develop liver diseases and concluded that this disease condition could be a major contributory factor to their illness and death.
Changes in serum gamma glutamyl transferase (γ-GT), carbohydrate deficient transferin (Wetterling et al., 1999), alanine aminotransferase, ALT, aspartate aminotransferase, AST (Wetterling et al., 1999; Onyesom et al., 2007) and bilirubin (Onyesom et al., 2007) have been used as laboratory markers to assess liver function in alcoholics. These measurements yielded elevated average levels, but none monitored the trend of these diagnostic analytes through the various stages and severity of liver disease or injury.
This study, determined the changes in serum bilirubin (total, conjugated and unconjugated), ALT and AST over a 15 week exposure to ethanol consumption. This design is therefore, capable of reporting the changes at the normal, early and late disease conditions of the liver induced by ethanol.
Experimental Animals
Eight adult male albino rabbits with an initial mean weight of 1.46 kg were purchased from a local farmer in Warri, Delta State, Nigeria. The rabbits were housed singly in clean metal hutches and acclimatized on growers’ mash (a product of Bendel Feeds and Flour Mills [BFFM] Ltd., Ewu, Nigeria) and laboratory condition, for 10 days before commencing the experiment. The rabbits were then divided into control (normal saline-treated) and test (ethanol-treated) groups with four animals each.
Feeding
The animals were given about 80g wet weight of the growers mash/kg body weight/day. The feeds were mixed with water in a ratio of 10:1 (w/v) in order to achieve a texture acceptable to the animals. Clean water was provided for the animals to drink ad libitum. Stale feed remnants were regularly discarded and the feeding experiment was conducted at room temperature (30±2°C).
Administration Procedure
The test-group animals were orally given 1.5 g (40%) ethanol/kg body as singly daily dose, while the control rabbits received the equivalent amount of normal saline in lieu of ethanol. Normal (or physiological) saline is a placebo used to control the administration and effects of ethanol. The ethanol dose and normal saline were administered after about 45 min of feeding the animals in the morning. The animals were exposed to these treatments along with their daily feeding for a regular period of 15 weeks. The study was conducted in the Alcohol Laboratory Unit, Department of Biochemistry, Delta State University, Abraka, Nigeria and the animal care complied with the guideline of the National Institutes of Health (NRC, 1985).
Blood Sample Collection
Fasting whole blood was collected from the vein in the right ear of the rabbits at the 5th, 10th and 15th week of commencing the treatments, into plain sterile tubes, using disposable, 21-guage hypodermic needles. Basal samples were also collected prior to commencement. The blood samples were then centrifuged at 1,200 x g for 5 min at room temperature to separate the sera samples which were stored frozen and analysed within 48 h of collection.
Laboratory Analyses of Samples
Serum bilirubin (total, conjugated and unconjugated) were determined by the diazo reaction method (Noslin, 1960). The activity values of AST and ALT in the serum samples were both estimated by colorimetric method (Reitman and Frankel, 1957).
Histopathological Examination
At the end of the 15-week exposure, the rabbits were sacrificed by cervical dislocation under anaesthesia and their livers were quickly excised and then, rinsed in cold normal saline and immediately fixed in 10% formol saline. They were in turn prepared for histopathological examination using the hematoxylin and eosin (H and E) staining technique, at the Department of Morbid Anatomy, University of Benin Teaching Hospital, Benin City, Edo State, Nigeria. The developed slides were examined and interpreted by a consultant pathologist.
Statistics
Statistical significance of the differences between treatment groups was assessed by Student t-test. The time-courses of serum analytes were analysed by repeat measure analysis of variance (ANOVA) followed by the Dunnett’s test for multiple comparisons and statistical significant difference was established at p<0.05. EPI computer software was used.
Table 1 shows changes in serum bilirubin (total, conjugated and unconjugated) obtained from the control (saline-treated) and test (ethanol-treated) animals at the 5th, 10th and 15th week of exposure. Changes in serum total bilirubin for the control animals were minimal (between 4.8-9.7% increase) but ethanol treatment induced a progressive higher levels ranging from 21% increase at the 5th week to 30.7% increase at the 15th week. Such observed increases in percentage mean differences from basal (0 week) value may have been caused by reduced hepatic conjugation as evidenced by the increased levels of unconjugated bilirubin (81-243%) and decreased amounts of conjugated bilirubin (10-29%) in serum.
Changes in pathophysiological enzyme activity values show that ethanol treatment respectively increased AST basal (0 week) value (18.8±3.8 IU L-1) to 24.2±4.3, 26.8±4.6 and 24.6±4.7 IU L-1 at the end of the 5th, 10th and 15th week of exposure. These values were 28.7% (p>0.05), 42.6% (p<0.05) and 30.9% (p>0.05) higher than the basal value. Similar comparison for the control (normal saline-treated) animals were observed to be 1.6% (p>0.05) at the 5th week, 1.1% (p>0.05) at the 10th week and 4.8% (p>0.05) at the 15th week of treatment.
Changes in ALT activity values were respectively increased by 14.1% (p>0.05), 28.8% (p<0.05) and 15.9% (p>0.05) at the end of the 5th, 10th and 15th week of exposing the animals to ethanol. Those of the control rabbits increased from 0.6 to 1.6% and then decreased to 0.3%.
| Table 1: | Changes in some diagnostic markers of liver function in rabbits following ethanol consumption |
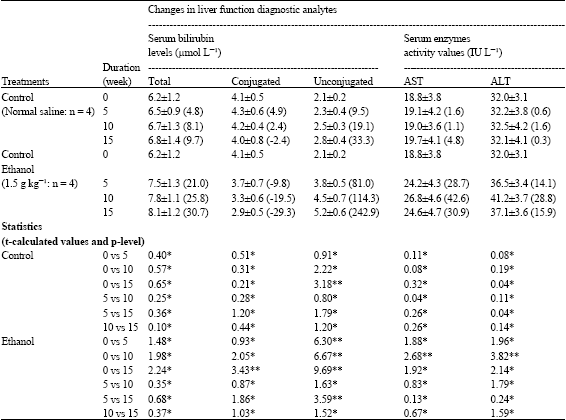 | |
| Values of the diagnostic analytes are expressed as Mean±SD of n rabbits. Values in parenthesis are percentage differences from basal (0 week) value. t-table = 2.35 at p = 0.05; *p>0.05; **p<0.05 | |
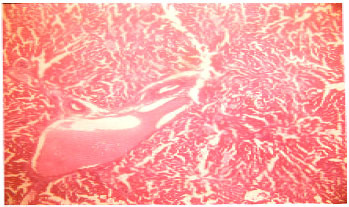 | |
| Fig. 1: | The liver tissue consisting of portal triad, central vein and plates of hepatocytes. Normal liver tissue (H and E Staining X 40) |
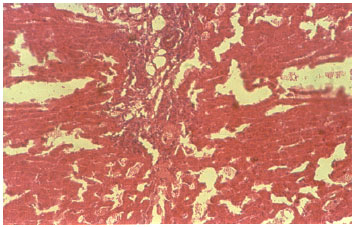 | |
| Fig. 2: | The liver shows periportal chronic inflammation infiltrate and mild periportal fibrosis and plates of hepatocytes. Fibrotic liver tissue (H and E Staining X 40) |
| Table 2: | Changes in conjugated:unconjugated bilirubin and AST:ALT ratios induced by ethanol administration in rabbits |
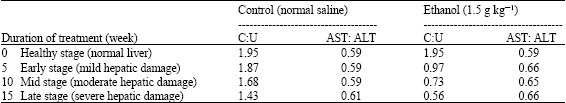 | |
Figure 1 and 2 show the microscopic changes in hepatic tissue for the control (Fig. 1) and ethanol-treated (Fig. 2 ) rabbits. The fibrotic changes seen in the hepatic tissue of ethanol-treated rabbits is indicative of alcohol induced fatty hepatitis and from this investigation, this disease condition is associated with a marked reduction in the ability of the liver to conjugate bilirubin and store AST and ALT.
The mean C:U and AST:ALT ratios for the control rabbits are 1.73±0.25 and 0.60±0.12, but at the 5th week of administering ethanol, these values became 0.97 and 0.66 (Table 2). At the 10th week of ethanol treatment, the C:U and AST:ALT ratios were observed to be 0.73 and 0.65 and these changed to 0.56 and 0.66 at the 15th week, the period at which fatty hepatitis manifested. Thus, these ratios vary with the degree of hepatic damage.
The effects of several hepatotoxic agents: carbon tetrachloride (Obi et al., 1998; Onyesom and Mordi, 2006), cyanide (Okolie and Osagie, 1999), ethanol (Reinke et al., 1988; Onyesom et al., 2007) on liver structure and function have been studied, possibly because of the liver’s central metabolic roles. The liver conjugates and excretes bilirubin, synthesizes ALT, stores AST and so, changes in serum levels of these analytes have become diagnostic tools and markers in assessing liver function.
The study demonstrates a progressive and marked increase (p<0.05) in the level of unconjugated bilirubin following ethanol administration (Table 1). This indicates an increasing loss in the ability of the liver to conjugate and excrete bilirubin. Onyesom et al. (2007) have observed that in alcoholics, the degree of bilirubin conjugation was reduced by about 28.9% when compared with control subjects. In this present study, conjugating ability was reduced by 29.3% after about 15 weeks of administering 1.5 g (40%) ethanol/kg to rabbits and this compares well with the human study.
Recall that the liver stores AST, but synthesizes ALT (Nduka, 1999). So, necrotic activity in the liver causes a release of abnormal quantities of AST and ALT enzymes into the blood. Treatment with ethanol induced a rapid increase in the serum activity of ALT to 14.1% at the 5th week and then to 28.8% at the 10th week, but was somewhat reduced to 15.9% at the 15th week. The AST percentage increase at these timed periods were 28.7, 42.6 and 30.9%, respectively (Table 1). AST activity was increased in a larger proportion. Ethanol (Onyesom et al., 2007), cyanide (Okolie and Osagie, 1999) and carbon tetrachloride (Obi et al., 1998; Onyesom and Mordi, 2006) administrations have been reported to increase serum AST and ALT activities, suggesting their hepatotoxic effects and AST activity is increased in a larger proportion in various types of liver disease, even though the organ is rich in ALT (Theal and Scott, 1996). This might be due to the cytoplasmic and mitochondrial origin of AST and this study appears to further confirm previous reports. Tremendous increase in the activity of serum ALT is primarily used as a test for hepatic dysfunction (Nduka, 1999) probably because it originates from the cytoplasm only.
The administration of 1.5 g (40%) ethanol/kg body weight to adult albino rabbits for a continuous period of 15 weeks caused fatty hepatitis as evidenced by the fibrotic changes in the microstructural appearance of the liver (Fig. 2). Wetterling et al. (1999) diagnosed fatty hepatitis in one-third of alcoholics sampled. Mordi (2006) observed an increased fat deposits by about 145.5% in the liver of Wistar rats treated with 1.5 g (40%) ethanol kg-1 for 7 days. Ethanol is almost entirely detoxified in the liver (Lieber, 1988), so hepatic disorders are common in cases of ethanol toxicity. The characteristic trend of the C:U and AST:ALT ratios (Table 2) and the pattern of the increases in AST and ALT activity values (Table 1) could be used to predict the stage and severity of hepatic disorder.